Wild-type
p53.mediated down-modulation
of interleukin
15 and
interleukin
15 receptors in human
rhabdomyosarcoma cells
C
DeGiovanni',
PNannil,
ASacchi2,
S
Soddu2,
IManni2,
GD'Orazi23,
SBulfone-Paus4,
TPohl4,
LLanduzzi15,
G
Nicoletti1-,
FFrabetti1,
IRossi' and P-LLoIlini'
1Carx
ResearchInsttute,
University
ofBoklona, Bologna,
italy MokcularOncogenesis
Laboratory,ReginaElenaCancerInstitute,Rome,Italy3Departbent
ofOncologyandNeuroscience,GD'AnnunzioUniversity,Chieti,Italy, InrttuteofImmunology,Berlin,Germany5ISTBiotechnoogySatelliteUnit,Boogna,ttaly
Summary We recentty reported thatrhabdomyosarcoma cell linesexpress andsecreteinterleukin 15(IL-15), atightty regulatedcytokine
with IL-2-like actvity.To testwhetherthe p53-impairedfunction that isfrequently found inthis tunour type couldplay a role intheIL-15 producbon,wild-type p53gene wastransducedin thehumanrhabdomyosarcoma cell line RD(whichharboursamutatedp53gene),and its
effectonproliferationandexpression of IL-15wasstudied. Arrestofproliferationwasinducedbywild-type p53:increasedproporionsof
G1-arrested cells andofapoptoticcells were observed. A markeddown-modulatin ofIL-15expression,atboththemRNA andproteinlevel,was
foundinp53-transduced cells.Becauseadirecteffect ofIL-15onnorrnalmuscle cellshas beenreported,thepresenceofIL-15membrane receptors was studied by cytofluorometnc
analysis.
Rhabdomyosarcoma cells showed IL-15 membrane receptors, which aredown-modulated bywild-type p53transfectedgene. In conclusion, wild-type p53 transductn in human rhabdomyosarcoma cells induces the
down-modulation ofbothIL-15productionandIL-15receptorexpression.
Keywords:p53; interleukin15; interleukin-15receptors; genetransducton; human rhabdomyosarcoma
Mutationinthe tumour-suppressorgenep53isthemostfrequent
alteration foundinhumantumours (Ko and Prives. 1996). Wild-typep53 is atranscription and translation regulator acting on a
varietyofgenesinvolved inthecontrol of cellcycle and differen-tiation (Ewen andMiller. 1996; Bourdonet al. 1997)thatcanbe either activatedor repressed. These genes includecytokines and growth factors (Fujiwaraetal. 1994;Shinetal. 1995: Peschetal.
1996:
Zhang
etal. 19%).Therefore. impairedp53
function could determine a deregulated production ofcytokines/growth factors.which could be of advantage for the malignant phenotype
(Baserga. 1994). Rhabdomyosarcoma. the most frequent soft-tissuesarcomainchildhood(Pappoetal. 1995).frequentlyshows
impaired p53function(Felixetal.
1992:
Dilleretal. 1995: Keletietal. 1996)andproducesavarietyofgrowthfactors (Schweigerer
et al. 1987; El-Badry etal. 1990: De Giovanni et al. 1995) that could be subject to transcription control byp53 (Fujiwara et al. 1994: Shinetal. 1995: Zhangetal. 1996). We recently found that
rhabdomyosarcoma cell lines express and secrete interleukin 15
(IL-15)(Lollinietal. 1997).Thiscytokine. controlled at both
tran-scriptionandtranslation levels.hasanIL-2-likeactivity (Tagaya et al. 1996;Onuetal. 1997)and protectsfromapoptosisboth in vitro and in vivo (Bulfone-Paus et al. 1997). IL-15 transcription has beenfoundinavarietyofnormalandneoplastic tissues (Grabstein
etal. 1994:Tagayaetal. 1996). but secretion of IL-15 protein is
restrictedto somenormal cell types andafew neoplastic celllines.
Received5December1997
Revised27May 1998 Accepted28May 1998
Correspondenceto:C DeGiovanni,Istituodi
Cancerologia,
VkaleFilopanti22,1-40126Bolgna,Italy
includingrhabdomyosarcoma (Lollinietal. 1997).Totestwhether impairedp53function couldplayaroleinIL- 15 production. we
transfectedp53geneinrhabdomyosarcomacells andstudied the effectsonproliferationandonIL- 15transcriptionandproduction.
Because cells of the muscle lineage can respond to exogenous IL-15 (Quinn et al. 1995). expression of IL-15 receptorswasalso studied.
MATERIALS
ANDMETHODS
Cells
andp53
genetransduction
Transduction ofp53 gene was perforned in the human rhabdo-myosarcomacell lineRD. which is knowntoharbour a mutation in codon 248 of thep53gene (Felix et al. 1992). Unless otherwise
specified, cells were cultured in Dulbecco's modified Eagle medium (DMEM) supplemented with 10% fetal bovine serum
(FBS)at370Cinahumidified 7%carbondioxide atmosphere.All media constituentswerepurchased fromLifeTechnologies. Milan. Italy. Plasmid pN53cG(Val-135). carrying the temperature-sensi-tivep53(ial-135) gene (ts-pS3)under thecontrol ofHaMSV-LTR
andthe selectable markerneounder the control of the RSV-LTR
(Sodduetal. 1994).andplasmidpRSVneo,carrying the selectable markeralone.were usedfortransfections. Approximately 3x 106
exponentially growing RD rhabdomyosarcoma cells were trans-fectedbyelectroporation(0.25 V. 960
jiF)
with a gene pulser (Bio-RadLaboratonres.
Hercules.CA. USA). Transfectants were clonedby limiting dilution in medium containing 0.75 mg mll G418
(Gibco BRL): one clone designated RD-tsp53 and one control
designated RD-Neowere thenstudied. All transduced cells were
routinelyculturedat37°C andshiftedtothepermissive
tempera-tureof32°Cforexperimentalstudies.
Toverifythetransactivatingactionofts-p53geneproductatthe
two culture temperatures, RD-Neo and RD-tsp53 transfectants were retransfected with a chloramphenicol acetyltransferase (CAT)reporter construct.Approximately 1 x105cellswereplated
in60-mm dishes and transfectedbycalcium
phosphate
precipita-tion withPG3CAT
vector,carryingtheCAT reporter genedrivenby a polyomavirus promoter and 13copiesof thep53
consensus-bindingsequence,or
MG,5CAT
vectorwith 15copiesofp53non-bindingsites(Kemetal, 1992)(kindly provided byBVogelstein).
Soon aftertransfection, the cellsweretrypsinizedand seeded in two new dishes, one of which wasincubated at the permissive
temperature and the other at thenon-permissivetemperature. After 48h,thecells were lysed by freezeand thaw. CATactivity was
determined usingthe two-phase fluorescence diffusion assay as describedpreviously(Huffetal, 1993);countsper minute(c.p.m.) were normalized to proteinquantity.
Overexpressionofwild-typep53wasalsoobtainedbyinfection
of RD cells with replication-defective, recombinant adenovirus carrying the human wild-type p53 cDNA (AdJL16), kindly
provided by Silvia Bacchetti (Bacchetti and Graham, 1993). AdherentRDcellswereinfectedwith 20p.f.u.ofAd-p53ord1312
control
adenovinus,
as described previously (Bacchetti andGraham, 1993). Forty-eight hours later, cells were subjected to
reversetranscnptase polymerasechain reaction(RT-PCR)analysis.
Proliferation studies
RD-tsp53and RD-Neo cellswereseeded in DMEM+10%FBSat
thedensity of 10 cells cm-2 and cultured at37°Cor32°C for 4
days,then cultures were switched toamedium withalow content of exogenous growth factors (DMEM + 2% horse serum) and incubation continued for further 7 days with medium renewal every2-3 days. At 3-4dayintervals,cultures wereharvested and cell number and viability were determined by trypan blue dye
exclusion test.
Forcellcycleanalysis,5x10 trypsinizedcells were washedin
PBS, fixedincoldmethanol-acetone (1:4)solutionfor 30mmat
4°C and DNA was stained with 50
ig
ml' propidium iodide(Sigma,St.Louis,MO, USA)in
phosphate-buffered
saline(PBS)with 2
jg
ml-' RNAase A (Boehringer-Mannheim Italia,Milan, Italy)for 30minatroomtemperature. Thecontentof DNA was measuredbyanEpics XLanalyser (CoulterCorporation,
Miami.FL, USA).
The proportionofapoptotic cells wasstudied on cellscultured
as above,harvested and stained with Hoechst 33342; about 500 cells were scoredforapoptoticbodies at 40xmagnification.
In someexperiments,cellcultures were treated with exogenous recombinant human IL-15 (Immunex, Seattle, WA, USA) at
concentrationsrangingfrom 100to 1000ng ml-, withanti-IL-15 monoclonalantibodyM11 (Immunex),at a finalconcentrationof
3
jig
ml-', or with anti-IL-15 polyclonal antibody (PeproTech, London,UK),atafinal concentration of 5jig
ml-';cellyield was determined4days later.IL-15
expression
Cells
transduced
and cultured as above were utilized forRNAextraction,cDNA synthesisand RT-PCR,asreported previously
(DeGiovanni etal, 1995;Lollinietal, 1997). RNA was extracted fromatleast three independent
experinments
in whichcontrol and25- 20-0 x E 15-0. 0 10
C:.)5
O--zzzn-I<9
%/
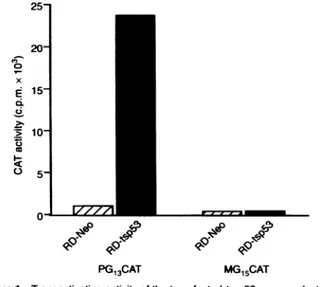
PG13CAT MG15CATFIgure 1
Transadivating actvity
of thebtansfectedts-p53ge produ atthe
permss
tenreratureof32°C.RD-Neo- andRD-tspb3-transfected
cellswererebtansectedwith
PG,3CAT
vector,carrying theCATreportergenedrivenbypolyofafirusprofoterand 13copes ofthep53
consensus-bdgsequenceorwiththecontrolMG,CATvectorwih15copiesofap53
non-biding
site.CATacivity(seeMaterialsandmethods) in cels cuituredat320C is shown
p53 transfectants were run inparallel; acDNApanel from each expernment was tested in RT-PCR at least twice. Specificprimer
pairs for glyceraldehyde-3-phosphate dehydrogenase (GAPDH)
andtransforming growth factor
0,
(TGF-3,)
wereobtained from Clontech (PaloAlto,CA. USA); prinmersfor IL-15 were synthe-sized as reportedpreviously(Lollini etal, 1997).Enzyme-inked
immunosorbent
assay(EUSA)
Cellswereseeded ataconcentrationof4-8x 104 cellscm-2and cultured at37°Cor32°C. After 4 days, cultures were switched to RPMI + 0.5% FBS andincubation continued for afurther4days at
37°C or at 32°C respectively. Cell supematants were then collected and IL-15 and
TGF-01
prductionmeasured by means of ELISA purchased from Genzyme (Milan, Italy) and usedaccordingtothemanufacturer's protocol. Supernatantsfor IL-15 determination were concentrated about 100 times as reported
previously
(Lollini
et al. 1997). At the sametime, cell cultureswereharvested and cell yield wasdetermined. Cytokine produc-tion was expressed in pg per 106 cells.
Cytofluorometic
sdies
Expression of membrane molecules on cells seeded at 106 cells cm-2 andcultured at 37°C or 32°C was detrmined by indirect
immunofluorescence and cytofluorometric analysis (FACScan, BectonDickinson, MountainView. CA, USA) using thefollowing primaryreagents: IL-15-IgG2b fusion protein, which recognizes all membrane IL-15 binding sites, produced by cells
trsduced
with ahuman IL-15-murine IgG2b fusion construct (Bulfone-Paus etal, 1997); monoclonal antibody M160(Immunex) recognizing thea-chain of the trimeric IL-15receptor, polyclonal C-16 anti-serum(Santa Cruz Biotechnology, Santa
Crz,
CA, USA)recog-nizing
TGF->-Rll;
anti-human class I major histocompatibilitycomplex W6/32 antibody (SeraLab, Crowley Down,
UK):
anti-CD44 monoclonalantibody.cloneIM7(PharMingen. San Diego.Britsh
Joumal ofCancer(1998)78(12), 1541-15464p
4pie
I(FIqA , RD-Neo RD-tsp3 a,.-//
/y
XRD-Neo
///i
X
3TC 37°C 32°C Adeno L0 to a 0 %-Z t Z 0 0 in 'cj 0 cc cc cc ECIT
3: IL-15 -TGF_ GAPDH 3 6 9 12 DaysofcultureFhgure2 Proliferationofts-p53genetansduced RD human
rhbdmyosarnacellscultured at37°C (mutated p53 conformation)and at
32°C (wiklype p53conotion). Open symbols,RD-Ne transfectnt
cells;closedsytbols,RD-tsp53tasea cels;dashed ine,37°C;
continuousline,32°C
CA, USA). The results shownarefromanexperiment
representa-tive ofatleasttwoindependent experiments. To study the
expres-sion of p53, the monoclonal antibody PAbl22 (Boehringer Mannheim, Germany) recognizing both wild-typeand mutatedp53
was used in indirect immunofluorescence on cells fLxed with
methanol.
RESULTS
Humanrhabdomyosarcoma cells RDwerewansduced with the
ts-p53gene,whoseproductbehaveslikewild-type p53atthe pennis-sivetemperatureof320C(Sodduetal, 1994). p53 expressionlevel
andtransactivating action at both permissive andnon-pennissive
temperatures were determined on transfectant cells. A high p53
expression was observedby cytofluorometry in RD-tsp53
trans-duced cellsatbothtemperaturesbymeansofanantibody that binds
top53 in the wildtypeaswell asin the mutated conformation(data
not shown). To verify the ransactivating action of ts-p53 gene product, RD-Neo and RD-tsp53 transfectants were retransfected
with PG
3CAT
vector, carrying the CATreporter genedriven by polyomavirus promoter and 13 copies of the p53consensus-bindingsequence orwith
MG,5CAT
vectorwith 15copies ofap53 non-binding site (Kernetal, 1992). Baseline levels of CAT activitywere observed in all transfectants cultured atthe non-permissive
temperature(datanotshown), whereasaveryhigh abilityto
trans-activate
PG13
drivenCAT expression was observed inRD-tspS3cellsculturedatthepermissivetemperature(Figure 1).
In RD-tsp53 transfectants, the induction of wild-type p53 conformationby cultureat32°C causedanalmostcompletearrest
ofcellgrowth(Figure 2); thesamecells culturedat37°C showeda
growth rate close to that of RD-Neo transfectant control cells. Proportions of
G,
cells and ofapoptotic cells were studied aspotential causesofgrowtharrestinducedby wild-type p53. The proportion of
G,
cells inRD-tsp53 transfectants culturedat32°C showedatwofoldincreaseoverthat ofRD-Neocontrols(72% and 36%, respectively) and a twofold increase in the proportion of apoptotic cells was also observed (3.9 ± 0.2% and 2.2 ± 0.2%respectively). B 10 0 10. 0. cm eg 5 IL-15 TGF-1 -0 40 10 a Go 0 Z f I * V - A _ 37°C 32WC 37°C 32WC
Figure3 Productionof IL-15and TGF-1 by p53genebansduced RDcels.
(A)ExpressionofmRNAforIL-15,
TGF-P,
andGAPDHevaluatedby RT-PCR. RNAwasextacted fromRD-NeoandRD-tsp53 transfectant cultures incbated4daysattheiricated teperature andfromadenovifectedcultures48 hafterinfection. ForeachRT-PCRprodud thefoling
ampnlficto cycles areshowntocomparecontrolandp53transfeant
samplesintheexponentialampifica phase: IL-15,30;
TGF-O,
25;GAPDH,20.(B) IL-15and
TGF-P,
releaseinsqermnaantsbyRD-NeoandRD-tsp53tansducedcellsculturedat37°Corat32WC
Theexpression of mRNA forIL-15wasstudied by RT-PCR in
comparison with the expression of an unrelated growth factor
(TGF-j1)
and ofahousekeepinggeneglyceraldehyde-3-phosphatedehydrogenase (GAPDH) (Figure 3A). Foreach primerpair, the
comparison between cDNAs derived from RD-Neo- and
RD-tsp53-transfectedcellswasmade in theexponential amplification phase. GAPDH showed that comparable cDNAs were obtained: PCR
products ofsimilar intensity were always observed after 15-25
amplification cycles. Adown-modulation of IL-15 tanscriptwas
observedinRD-tsp53-transfected cellsat370C andat320C(Figure
3A). The mean decrease in the intensity of ethidium
bromide-stainedbands, evaluatedby densitometricanalysis of five indepen-dentexperiments, was: 38.4% ± 8.6 at370C and 71.0%± 7.3at
32°C. Therefore, p53 down-modulation of 1L-15wassignificantly
higher (P=0.016) in the wild-type confonnation.
Down-modula-tion of the IL-15 transcript was constantly observed at different timepoints (e.g. from 2to 12days) after cell seeding.
Theexpression ofwild-type p53 proteinwasalso induced in RD
cells by infection withanadenovirus vector.againadecrease in
the]11-15 RT-PCRproductwasobserved inAd-p53 infected cells
in comparison with cells infected with the control adenoviral
BrtshJoumalofCancer(1998) 78(12), 1541-1546 1000- 10O-E 0 0 .o0 0 0 3. 0 10-Si I 0 e RD-tsp.
IL-15bindKg IL-1SRa
37C
TGF§-R
RD-NeoRD-tsp53
RD-NeoRD4sp53
Fgure4 Cytofluorotic analyss of membrane receptors for IL-15and
TGF-1 inRD-Neoand RD-tsp53transducedcellsculturedat370Corat
32°C. Ineachgraph,theabscissarepresentsfluorescenceintensityona
kogarithmicscalerangingfrom10°to10',andtheordinaterepresentsthe numberof cells.Open profiles,cells stained withsecondaryantbodyalone; shadedprofiles,cells stainedwithantbodyorfusionproteinrecognizing the indicatedspecficity
vector(Figure 3A). Meandecrease ± s.e.from three expenrments
was55.0%± 14.0.
Down-modulation of IL- 15 mRNA was not duetoan
unspecifi-cally
decreased transcription activity because the expression ofTGF-01
(Figure 3A)
wasslightlyincreased.Post-transcriptional
regulationof IL-15 protein production has beenreported (Tagaya
etal. 1996;Onuetal, 1997).Therefore, theexpression
of secretedIL-15protein wasstudiedbyanELISA in supematants from RD-Neo andRD-tsp53transfected cells (Figure3B).
AstrongdecreaseinIL-15productionwasalreadyobserved inRD-tsp53-transfected
cells cultured at 370Cand reached90%reduction in
32°C
cultures. Thisdecrease isunlikelytobedue to anon-specific
decrease in metabolicactivitybecausetheproduction ofTGF-13,
whose baseline level was approximately 200-foldhigher
than thatof IL-15. was actuallyincreasedunder the sameexperimental
conditions(Figure
3B).IL-15 binds to a membrane trimeric receptor. composed of a
specific
a-chain(IL-15Ra)
andof >- andy-chainsshared withotherinterleukins. Moreover,anunknown receptor has also beenreported onmastcells
(Tagaya
etal. 1996). ThepresenceofIL-15receptorson p53-transduced RD cells was studied by means of
cytofluori-metry.
eitherusingamonoclonalantibody recognizingIL-15Raor theIL-15-IgG2b
fusion protein (Bulfone-Paus et al. 1997) that detects cellmembranebindingsites.RD-Neo cellsexpressedIL-15membrane receptors.asshownbythebindingof the fusion protein. andatleastpart of thebindingwasdue to thepresenceofIL-l5Ra
(Figure 4).
Down-modulationofbothtotalIL-15membranebindingand ofIL-15Rawasobserved in
RD-tsp53-transfected
cells when thewild-type
p53conformationwasinducedbyculture at32°C. No differencewasobserved forTGF_j-RHg
(Figure 4).ThetreatmentofRD-NeoandRD-tsp53 cells withexogenous recombinant humanIL- 15atconcentrationsupto 1000ng ml-'. as well as with either monoclonal or polyclonal anti-IL-15
anti-bodies,
didnotdetermine anychangeincellproliferation and in expression of membraneglycoproteinssuch as HLA-A. B. C and CD44(data
notshown).
DISCUSSION
Humanrhabdomyosarcomacell line RDwas transduced with
p53
gene using twodifferent vector systems: ats-p53 plasmid
(Soddu
etal. 1994).giving riseto stable transfectants inwhichwild-type
p53 conformation isdependent uponthe culturetemperature. and
an adenoviral vectorcarrying the wild-type p53 gene (Bacchetti
andGraham. 1993). inwhich ahightransientwild-typep53gene
expressioncould be studied withinafew days afterinfection.With
bothvectorsystemsadown-modulation of IL- 15transcriptionwas
observed. Thiswasnotduetoanunspecificdecrease in
transcrip-tion activity because expression of an unrelated growth factor
(TGF-,B1)
wasslightly increased.In thets-p53system.down-modulationsof IL-15transcriptionand productionwerealso observedatthenon-pennissiveculture
temper-ature. but the effect was significantly higher when the wild-type
conformation was induced The effect of ts-p53 at
37°C
could be related to itsleakiness leading to alowamountof p53 in thewild-type conformationeven atthis temperature. However. experiments
performed at a
higher
temperature (390C) confirmed that thedecrease in IL-15
production
wasalready observed withts-p53in themutated conformation (data not shown). This suggests that both
mutated andwild-typep53wereactive in down-modulating IL-15. A
similar phenomenonhas been reportedfor other cytokinesin
expen-mental models basedonp53mutants. e.g. IL-2 andIL4are
down-modulated by wild-typep53aswellasby deletionmutantp53(Pesch
etal. 1996). TGF-j31
production
wasreportedtobeincreased byboth wild-type and mutated p53 inaglioblastomacell line (Fujiwaraetal.1994): this finding was extended to rhabdomyosarcoma by the
presentstudy withtemperature-sensitivep53geneproducts.
Although the above-mentioned IL- 15 down-modulation was
induced by bothwild-typeand mutatedp53. other effectsreported herewerespecific for p53inthewild-type conformation. suchas
the growth arrest and the down-modulation ofIL-15 receptors.
These different behaviours could suggest that different
mecha-nismsofp53actionareinvolved.
The presence of twop53 consensus sequences (the decamer
PuPuPuCAf[T/APyPyPy)inthepromoterand/orintronicregions
of transactivatedgenesisamechanismforp53actionas
transcrip-tionfactor(Bourdonetal. 1997).IL-15promoterhasnot yetbeen
studied.butap53consensussequenceispresentinthefirstintron
of the IL-15 gene (GAACTGGCCT. position 18-27 of the
sequenceX91233of GenBank).
An altemative to p53 consensus sequence binding. a
'non-specific' mechanism basedonp53 proteininteraction with general
transcription factors(Ko and Prives. 1996). has been hypothesized
forp53-induceddown-modulationofIL-2 and IL-4 (Peschetal.
1996). IL-15could share this mechanismwith otherinterleukins.
Itmustbe remembered that IL-2 and IL-15 share receptors and
activities (Grabsteinetal. 1994;Tagayaetal. 1996).
Different p53 propertiesarerelatedtodifferentproteindomains
(Koand Prives. 1996). The ts-p53 gene wetransfectedharbours
the temperature-sensitive Val-135 mutation in the DNA-binding
centralcoreregion: alterations inthis domainareresponsiblefor
properties ofp53 leading to increased tumorigenic potential of
cellsresulting fromchanges in the abilitytobindDNA. Val-
135-mutatedp53. however,couldmaintain, atleast inpart theability
to interact with general transcriptionfactors through the
amino-terminal domain.andthis could leadtothe transcription modula-tion by mutated p53found forsomecytokines(IL-2/4 and IL- 15) (Peschetal. 1996.presentstudy).
It is also possible that some of the reported effects may be related to the stateof the cell cyclemachinery. not directly viap53 protein-protein interactions. The study of p21 --FI. a key down-stream effector of p53 inhibition of cell cycle, will helpin clari-fying which mechanism is involved in different effects. P211lF'
transfection (Meng et al. 1998) as well as treatment with
exoge-nous p2l--'F peptides (Ball et al. 1996) could mimicp53 gene effects related to the state of cell cycle machinery. If p21w-F does not mimicp53 effects. thenitis more likely that the mechanismis
related directly top53 non-specific suppression oftranscnrption.
Afew IL- 15transcription modulators [e.g. UV radiations and y-interferon(IFN-y)] which leadtoanup-modulationinnormal cells
areknown so far (Tagayaet al. 1996). Wild-type p53 leadsto a
decreased transcription: this is the first example of a down-modulationof IL-15.
Thestudyof proliferative ability of stableRD-tsp53transfectants showed that whereas at non-permissive temperature
RD-tspS3-transduced cells had a
growth
curve close to RD-Neo control. expression ofp53inthewild-type conformation ledto analmost complete arrestof growth that could be determined by increaseinGI
arrestandin apoptosisrate. IL-15protectsfromapoptosis both invitro and invivo (Bulfone-Paus et al. 1997). Therefore. IL-15andp53appearto exert
antagonistic
rolesoncell apoptosisrate.IL- 15 produced by rhabdomyosarcoma cells could act in a
paracrine wayin the tumourmicroenvironment. interacting with cells
expressing
IL-15 receptors. such as lymphocytes and mastcells. butatpresentit is notknown whether andhow this cytokine modifies the tumour-hostrelationship. Moreover. IL-15 has been reportedtopromoteangiogenesisin vivo(Angiolilloetal.1997).a
phenomenon thatisbecomingmoreandmorerelevantfor malig-nancy. IL-15 release in culture medium by rhabdomyosarcoma cellsreached very lowlevels.of the orderof 5-10pgml-' (present data and Lollini et al. 1997). These levels areapparently lower than thethreshold(about 25
pg
mn-' )requiredforin vitrolympho-cyte proliferative responses (Grabstein et al. 1994: our
unpub-lished data). butinthelocalinvivo microenvironmentshort-range
acting
cytokinescould have significant effectsevenatlowconcen-trations.Inparticular.forIL- 15ithas beensuggestedthat
receptor-positive cells able to release the cytokine could store a high
concentration of IL-15 molecules on their surface. ready to be
presentedtopassengercells(Meazzaetal. 1997).
Thepossibility that IL- 15 alsoacts in anautocrinewaymustbe considered because RD rhabdomyosarcoma cellsdisplayboth the
specific a-chain of the trimeric receptor and IL-15 membrane
binding,
sites.asshownbytheIL-15-IgG2bfusionprotein. Some data on the effectiveness of IL-15 in muscle cells have been reported(Quinn et al. 1995). We arecurrently studying whetherautocnne effects (suchas induction ofproliferation or modifica-tionsinthe expressionofmembrane molecules) are determined by IL-15inrhabdomyosarcoma cells. However.in vitrotreatmentof rhabdomyosarcoma cells with exogenous recombinant IL-5 or
withmonoclonal and polyclonal anti-IL-15 neutralizingantibodies did not determine any modification in cell proliferation and in
expressionof membraneglycoproteins suchasHLA-A. B.C and CD44. This is in accordance with the absence of proliferating
effects observed in normal muscle cells
(Quinn
etal. 1995). In conclusion. aselective down-modulation of both IL-15 and IL-15 receptorswas found inrhabdomyosarcoma cells transduced withwild-typep53. This indicatesacontrolof IL-15circuitsbyp53andsuggeststhatfurther efforts should be directedtothestudy of the role of IL-
15S
in theprogression of human rhabdomyosarcoma.NOTE ADDED
INPROOF
The recent availability of the IL-15 promoter sequence (reported
byAzimi et al. 1998. Proc Natl Acad Sci USA 95:2452)prompted us toreanalyse all available IL-15 sequences forp53 consensus binding sites. No consensus sequence was found in the IL- 15 promoter.whereas several clusters of one. two and four decamers (with 0. 2 and 4 mismatchtolerance respectively) were found in introns3. 4 and 5 and in the last exon(GenBankX91233).
ACKNOWLEDGEMENTS
This work is dedicated to thememory ofProfessor
Giorgio
Prodi. MD. PhD. 10 yearsafter his untimely death. The authors would like to thank Gabriella Madriali for her excellent secretarial assistanceand Dr T Troutt (Immunex)forproviding recombinant human IL-15 and monoclonal antibodies Ml 11 and Ml 60. This work was supported by grants from National Research Council. Associazione Italiana per la Ricerca sul Cancro and MinisterodellFUniversit,aedella RicercaScientificaeTecnologica. Italy. IR is in receiptof a Fellowship from the AIRC.
REFERENCES
AngiolilloAL.KaneganeH.SgadariC. Reaman GH and Tosato G 1997i
Interleukin-15 promotesangiog-enesisin vivo. BiochemBiophYsRes Commun 233: 231-237
Bacchetti S andGraham F 1993ihnhibition of cell proliferationb%adenovirus vectorexpressingthehumanwild type p53 protein IntJ Oncol3: 781-788 Ball KL Lain S. Fahraeus R.Smvthe C and Lane DP(1996 Cellcvclearrest and
inhibitionofCdk4 activitv by small peptides based on thecarbox%--terminal domain ofp2l "> CurrBiol7:71-80
BasergaR 1994( Oncoggenes and the strategN ofgrowthfactors. Cell 79: 927-930 Bourdon J-C.Deg-uin-ChambonV.Lelong J-C. DessenP.Ma P. Debuire Band
Mav E (1997 Furthercharacterization of thep53responsiveelement
-identification of new candidategenes for trans-activation b% p53. Oncogene 14: 85-94
Bulfone-Paus S.Ung-ureanuD.PohlT. LindnerG.PausR.RuckertR.Krause Hand Kunzendorf U(1997( Interieukin-15 protects from lethalapoptosisin -%'o. .Vature Med 3: 1124-1128
DeGiovanni C. Melani C.NanniP. LanduzziL. Nicoletti G. Frabetti F.Gnrffoni C. Colombo MP and Lollini PL(1995 Redundancy ofautocrineloopsinhuman rhabdom%-osarcoma cells. Induction ofdifferentiationb'-suramin. BrJCancer 72:1224-1229
Diller LSexsmithEGottliebA. LiFP andMalkin D(1995 Germlinep53
mutationsarefrequently detectedinyoungchildrenwith rhabdomyosarcoma-JClinInvest95: 1606-1611
El-Badr OM.MtinnitiC. Kohn EC.HoughtonPJ.Daughada% WAHand HelmanLI
(1990 Insulin-likegrowthfactor II actsasanautocrine growth and motlitv factor inhuman rhabdomvosarcoma tumors. Cell Growth Different 1: 325-331 EwenMEandMillerSJ(1996(p53andtranslational control.BiochimBiophvsActa
1242:181-184
Felix CA.Chavez Kappel C.MitsudomjT. Nau MM. TsokosM1.Crouch GD. Nisen PD.WinickNJand HelmanLI 1992' Frequency and diversity of p53 mutationsinchildhood rhabdomr osarcomaCancerRes52: 2243--2247
FujiwaraT.Mukhopadhvax T. CaiDW.Morris DK. Roth JA andGrimm EA(1994(
Retroviral-mediatedtransductionof p53 gene increases TGF-beta expression in
ahumanglioblastomacell fine.IntJ Cancer56:834-839
Grabstein KH. Eisenman J. Shanebeck K_ Rauch C. SrinivasanS.Fung\V Beers C.
RichardsonJ.SchoenbornMA. Ahdieh M. JohnsonLAlderson AR. Watson
JD. Anderson DM andGiri JG(1994)CloningofaTcellgrowth factorthat interacts withthebeta chainoftheinterleukin-2 receptor. Science 264: 965-968
HuffCA. YuspaSH andRosenthalD( 1993( Identification ofcontrol elements3'to the human keratin Igenethatregulatecell tpeanddifferentiation-specific expression.J BiolChem268: 377-384
Keleti J.QuezadoMM. Abaza WM. RaffeldM andTsokosMi(1996(TheMDM2 oncoproteinisoverexpressedinrhabdomrnosarcomacell lines and stabilizes
wild-typep53protein.AmJPathol 149:143-151
Kern SEPietenpol JA. Thiagalingam S. SeymourA. Kinzler KW and Vogelstein B 1992)Oncogenicformsof p53 inhibitp53-regulatedgene expression. Science 256:827-830
KoLU andPrives C (1996) p53: puzzle and paradigm. Genes Dev 10: 1054-1072
Lollini P-L Palhneri G. De Giovanni C.Landuzzi L Nicoletti G. Rossi I.Gnrffoni C. Frabetti F. ScotlandiK.Benini S. Baldini N. SantoniA andNanni P 1997) Expression ofinterleukin-15(IL- 15)inhumanrhabdomyosarcona. osteosarcoma- and Ewing's sarcoma Int J Cancer 71: 732-736
Meazza R.GaggeroA.NegliaF. Basso S. Sforzini S. Pereno R.AzzaroneB and FerriniS 1997) Expressionof tw-ointerleukin- 15 mRNAisoformsin human
tumorsdoesnotcorrelate withsecretion: role of different sigmalpeptides. Eur J Immnunol 27: 1049-1 054
MengRD. Shih H.PrabhuNS. GeorgeDLandEI-Deirn WS (1998)Bypassof abnormalMDM2 inhibition of p53-dependent growthsuppression. Clin Cancer
Res4:251-259
Onu A.Pohl T. Krause H and Bulfone-Paus S (1997)Regulationof IL-15 secretion via the leader peptide of two IL- 15 isoforms. JImmunol 158: 255-262
PappoAS.ShapiroDN. Cn'st WM andMaurerH-M (1995)Biolog- andtherapyof pediatncrhabdomr osarcoma J Clin Oncol 13: 2123-2139
PeschJ. Brehm U. Staib C and Grummt F(1996)Repressionofinterleukin-2 and interleukin-4 promoters b- tumorsuppressorproteinp53. J InterferonCr-tokine
Res 16:595-600
QuinnLS.HaugkKLandGrabsteinKHR 1995)Interleukin-1i:anovelanabolic c,vtokine for skeletal muscle.Endohrinology136: 3669-3672
SchweigererL Neufeld G.MergiaA.Abraham JA.Fiddes JC andGospodarow-icz
D(1987)Basicfibroblast growthfactorin humanrhabdomyosarcoma cells: implications fortheproliferationandneovascularization of myoblast-derived tumors.ProcNail Acad Sci LISA 84:842-846
ShinTH. PatersonAMand KudlowJE (1995)p53 stimulates transcription fromthe
humantransforminggrowthfactor alpha promoterapotential growth-stimulatorn role for p53. Mol Cell Biol 15: 4694-4701
SodduS. Blandino G. Citro G. ScardigliR. Piae.eio G.Ferber A. Calabretta Band SacchiA(1994)Wild-tvpe p53geneexpressioninduces granulocytic differentiation of HL-60 cells.Blood 83: 2230-2237
Tagaya Y.Bamford RN.Defilippis AP andWaldmann TA(1996%) L- 15: a pleiotropic cytokinewithdiversereceptor/signalingpathways whose expressioniscontrolledatmultiple levels.Immunity4: 329-336
Zhang L. Kashanchi F. Zhan Q. Zhan S. Brady IN. Fornace AJ. Seth P and Helman Ui (1996)Regulationof insulin-likegrowthfactor 11 P3 promoterbyp53:a
potential mechanism fortumori2enesis.Cancer Res56: 1367-1373