Gut1993;34:1370-1373
Abnormal
plasma
polyunsaturated fatty
acid
pattern
in
non-active
inflammatory
bowel
disease
MEsteve-Comas,MCNunfez,FFernaindez-Baniares,AAbad-Lacruz,AGil,ECabre, FGonzailez-Huix, X Bertran, M A Gassull
Departmentof
Gastroenterology, HospitalUniversitari Germans TriasiPujol, Badalona, Spain MEsteve-Comas FFernandez-Bafiares AAbad-Lacruz ECabre FGonzilez-Huix XBertran M AGassull ResearchDepartmentof UNIASA,Granada, Spain MCNuinez A Gil Correspondenceto:
Dr MAGassull,Head of the
Department of
Gastroenterology, Hospital
Universitari'Germans Triasi
Pujol',CarreteradelCanyet
s/n, 08916Badalona, Spain. Acceptedforpublication
9February1993
Abstract
An abnormal plasma polyunsaturated fatty
acid pattern (PUFA) (increased n3 and de-creased n6PUFA) has been reported in active inflammatory boweldisease(IBD).The possi-bility ofaprimary defectinthe PUFA meta-bolism in IBD washypothesised. The aimof thisstudywastoassessplasmaPUFApattern
in inactiveinflammatoryboweldisease andto
ascertain whether patients who had had a
colectomyandwhoweresufferingfrom ulcer-ative colitishaveasimilarPUFApatternthan
those patients with non-active ulcerative
colitis and who had not had a colectomy.
Plasma fatty acids were analysed by
semi-capillarycolumngas-liquidchromatographyin
threegroupsofpatients with inactive IBD (24
patients with inactive ulcerative colitis who
had not had a colectomy, 15 patients with
ulcerative colitis who had had a colectomy, and 27patientswithCrohn'sdisease).Plasma
concentrationandpercentage ofC22:6n3 and
unsaturationindexweresignificantlyhigherin
patientswithinactiveulcerative colitiswithout a colectomy and the Crohn's disease group
(p<0O0001) than in controls. Plasma concen-tration and percentage of C22:6n3 and the unsaturation index remained significantly
higher,inboth the operated and non-operated ulcerative colitispatientswhencomparedwith controls (p<00001). These results suggest
that ininactiveIBD,anincreased PUFA
bio-synthesismightbethecauseof thehighvalues
of n3 compounds. These findings although
seenin activedisease, aremorenoticeable in
remission because ofthe lack ofartefactual
factors(malnutrition, steroids, inflammation).
Inaddition, persistenceofhighvalues in both
groups ofulcerative colitis patients-thatis,
thosewho had hadacolectomyandthose who had not suggests the existence of a primary
abnormalityin thePUFAmetabolism in IBD. (Gut1993;34:1370-1373)
Polyunsaturated fattyacids(PUFA) participate
in membrane function,'2 including that ofthe
immune cells34 and they are precursors ofthe eicosanoids.'-8 For thesereasons,theymayplay animportantpartinthepathogenesisof
inflam-matorybowel disease(IBD).
We have described an abnormal PUFA
pat-tern in active IBD.9 This consists ofincreased values of the precursor (C18:3n3) and the end product(C22;6n3)of the n3 seriesanddecreased values ofdihommo-y-linolenicacid(C20:3n6).A
stepwise decrease in PUFA values, which was
morenoticeableforthen6serieswasalsoseen as
the disease becamemoresevere. Eveninsevere
disease,however, plasmaconcentrations of the
n3PUFA remainedhigherthan thosein
healthy
controls. These findingssuggested that in IBD there might be a primary increase in PUFA
biosynthesis. In active IBD this phenomenon
wouldbepartly counterbalanced byanincreased fatty acid utilisation because of factors associated with disease activity, such as inflammation,
hypermetabolism, malnutrition, and steroid
treatment.
Onthe basis of the abovefindings, it could be
hypothesisedthat in the absence of otherfactors,
that isin inactivedisease, the values of long chain PUFA would be even higher. Therefore, the study of patients with inactive IBD would be of interest. In addition, to ascertain the possible existence ofa primary defect in PUFA
meta-bolism, the study of patients with ulcerative colitis who had had a colectomy would be
particularlyimportant, as in such patients, the
target organhas been removed. The aim of this study was to assess prospectively the plasma fatty acidpatternin patientswith inactive IBD (both ulcerative colitis and Crohn's disease),and
in ulcerativecolitis patients aftercolectomy.
Patients and methods
PATIENTS
Threegroups of66patients with inactive IBD
wereincluded in the study: 27 Crohn's disease (16 men, 11women;medianage24years;ranges
14-78 years), 24 ulcerative colitis patients (10 men, 14 women; median age 41 years; range: 23-75 years) and 15 patients with ulcerative colitis who had had a colectomy, four
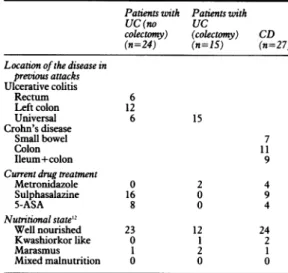
procto-TABLEI Clinicalfeatures ofthe patients
Patients with Patients with
UC (no UC
colectomy) (colectomy) CD (n=24) (n=15) (n=27) Locationof the disease in
previous attacks Ulcerative colitis Rectum 6 Left colon 12 Universal 6 15 Crohn's disease Small bowel 7 Colon 11 Ileum+colon 9
Currentdrug treatment
Metronidazole 0 2 4 Sulphasalazine 16 0 9 5-ASA 8 0 4 Nutritionalstate'2 Well nourished 23 12 24 Kwashiorkor like 0 1 2 Marasmus 1 2 1 Mixed malnutrition 0 0 0
UC=ulcerativecolitis, CD=Crohn'sdisease. 1370
Abnormal plasmapolyunsaturatedfatty acid pattern in non-active inflammatory boweldisease
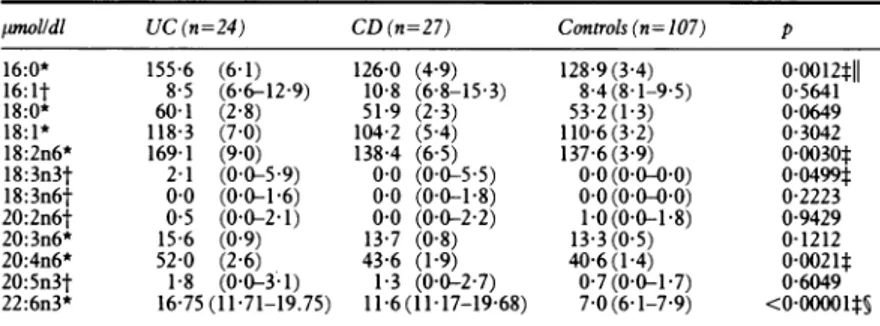
TABLE II Plasmafatty acids in inactiveinflammatory bowel disease
pnolldl UC(n=24) CD (n=27) Controls (n= 107) p 16:0* 155 6 (6-1) 126-0 (4 9) 128-9(3 4) 0-0012tll 16:1t 8 5 (6-6-12-9) 10-8 (6-8-15-3) 8-4(8-1-9-5) 0-5641 18:0* 60-1 (2 8) 51-9 (2 3) 53 2(1-3) 0-0649 18:1* 118-3 (7 0) 104-2 (5 4) 110-6(3 2) 0-3042 18:2n6* 169-1 (9-0) 138-4 (6 5) 137-6(3 9) 0 0030t 18:3n3t 2-1 (0 0-5 9) 0-0 (0 0-5-5) 0 0 (0 0-0 0) 00499t 18:3n6t 0 0 (0-0-1-6) 00 (0-0-1-8) 0 0 (0 0-0 0) 0-2223 20:2n6t 05 (0-0-2-1) 00 (0 0-2-2) 1-0(0 0-1 8) 0-9429 20:3n6* 15 6 (0 9) 13 7 (0 8) 13 3 (0 5) 0-1212 20:4n6* 52 0 (2 6) 43-6 (1-9) 40-6(1-4) 0-0021t 20:5n3t 1-8 (0-0-3;1) 1-3 (0 0-2-7) 0-7(0-0-1-7) 0-6049 22:6n3* 16-75(11-71-19.75) 11 6(11 17-1968) 7-0(6-1-7-9) <0o00001ts
*Mean(SEM). One way ANOVA+Scheffe test; tmedian (95% CI), Kruskall-Wallis test+Mann-Whitney U test; tUC vcontrols; §CDvcontrols; ||CDvUC. Otherabbreviations as in Table I.
TABLEIII Plasmafatty acidsininactive inflammatory bowel disease
% UC(n=24) CD (n=27) Controls (n= 107) p 16:0* 25-7 (0-3) 24-3 (0 4) 25-2 (0-2) 0-0515 16:It 14(0 9-2 1) 22(1 3-3 1) 1 7(1 5-1 9) 0 1456 18:0* 9-9(0-2) 10-0(0 3) 10-5 (0-1) 0-0811 18:1* 194(0-7) 20-0(0 6) 21-7(0-3) 0-0061t 18:2n6* 27-8 (0 9) 26-9 (1-0) 27-2 (0 4) 0-7817 18:3n3t 0 3(0-0-1-0) 00(0 0-0 9) 0 0 (0 0-0 0) 0-0815 18:3n6t 0 0(0 0-0 2) 00 (0-0-0-3) 0 0 (0 0-0 0) 0-2480 20:2n6t 0-1(0-0-0-3) 00(0 0-0-4) 0-1(0-0-0-3) 0-5297 20:3n6* 2-5(0-1) 26(0 1) 2-6(0 1) 0-9068 20:4n6* 8-6(0-3) 8-4(0-3) 8-0 (0-2) 0-4427 20:5n3t 0-2(0 0-0-5) 0-2(0 0-0 6) 0-2(0-0-0-3) 0-8298 22:6n3t 2-6(1-9-3.5) 2-4(2 3-3 3) 1-4(1-3-1-6) <0-00001t§ UNID* 141-1(1-7) 140-7(1-8) 131-5(1 1) 0-00010t§
*Mean(SEM). One way ANOVA+Scheffe test; tmedian (95% CI), Kruskall-Wallis test+Mann-Whitney U test;tUCvcontrols; §CDvcontrols.UNID=unsaturation index. Other abbreviations as
inTableI.
colectomy and 11 colectomy plus ileal pelvic pouch (eight men, seven women; median age 31 years, range: 15-46 years). Patients were
considered inactive asassessed bytheTruelove and Witts' index'" for ulcerative colitis and
the Van Hess' index" for Crohn's disease. No patient with ileal pelvic pouch had clinical, endoscopic, or histological signs of pouchitis.
Table I shows the clinicalfeatures ofthe patients
studied.12The resultsofthefatty acidstateof all ofthesepatients hadbeenreportedpreviouslyin
activephase.9
CONTROLS
Apreviouslyreportedgroupof107healthy,well
nourishedsubjects (46men, 61 women;median
age: 32 5 years; ranges 18-76years) acted as a controlgroup.9
Informed consent was obtained from each patient and healthy controls. The study was
performed in conformance with the 1975
Declaration of Helsinki ethical guidelines and was approved by the Research and Ethical
Committees of theHospital.
PLASMA FATTY ACID ASSAY
Inallpatientsandhealthycontrols,a5 mlvenous bloodsampleforplasmafattyacidmeasurement wastakenafter a 14hourovernightfast. Blood
samples in all ulcerative colitis and Crohn's disease patients were obtained three months after they had been considered inactive and in ulcerative colitis patients who had had a colectomy between three to six months after completionof alloperations. Themethods used
for plasmafattyacid assay have beendescribed in
detail elsewhere.91' " Heptadecanoic acid
(C17:0) was added as an internal standard to allow fatty acid concentrations in plasma total lipids to be determined as absolute values, and notonly as percentages.
Fattyacidfrom14:0 to22:6n3weremeasured.
Unidentified peaks accounted forless than 0 5% of the totalfatty acid. Resultsareexpressedas a
total fatty acid concentration
([tmol/dl)
and as apercentage distribution of each fatty acid. Theunsaturation index (UNID)wascalculated accordingtotheformula'5:UNID=> (fatty acidpercentagex numberofdouble bonds).
STATISTICAL ANALYSIS
For statistical analysis the Statistical Package for
Social Sciences SPSS/PC+ (SPSSInc,Chicago,
Illinois, 1985) wasused.'6Variables with normal
distribution and homogeneous variance were
compared bymeans ofparametric tests,
other-wise their non-parametric counterparts were
used. One way analysis of variance with 'a posterior' Scheffe test or Kruskall-Wallis one way analysis of variance by ranks were used.
When the Kruskall-Wallis test disclosed a
significant p value, the Mann-Whitney U test wascarried out to detect where thedifferences occurred. Resultsareexpressedasmean(SEM)
or median and the 95% confidence intervals
(CI)'7 for parametric and non-parametric variables respectively. Because agedistribution
wasdifferentamongthegroupsstudied and this
isafactorinfluencing plasmalipids,'8theresults
were adjusted forthe effect ofage bymeans of
analysis
of varianceusing
ageas acovariate.Results
PLASMA FATTY ACIDS ININACTIVE INFLAMMATORY BOWELDISEASE
Tables II andIII detailrespectivelythe plasma fatty acid concentration and the percentages of
thedifferentfattyacidsinpatientswith inactive ulcerative colitisandCrohn'sdisease.
Themoststrikingfinding intheplasmafatty
acidprofile in inactiveIBDwastheconsiderable
increaseof docosahexaenoicacid(C22:6n3),the
finalproduct of then3 series,both inulcerative
colitis andCrohn'sdisease whencomparedwith
controls. This fact was seen when the results
wereexpressed eitherasabsolute concentration
or as apercentage.
Arachidonic acid(C20:4n6),themainproduct of the n6 series, showed the same tendency.
Statistical significancewas onlyobtained, how-ever, between the ulcerative colitispatientsand
controls, when the values were expressed as
absoluteplasmaconcentration. Absoluteplasma
values of linoleic acid (C18:2n6), the essential precursorof the n6 series, and ct-linolenic acid
(C18:3n3), the essential precursor of the n3
series, were also increased in ulcerative colitis
(TableII).
Asaconsequence of the increase inlongchain andhighlyunsaturatedPUFA,thevaluesof the
Esteve-Comas, Nuiniez, Ferndndez-Bainares,Abad-Lacruz, Gil,Cabre, Gonzdlez-Huix,Bertrdn,Gassull
TABLEIV Plasmafattyacids in inactive ulcerative colitis. Influence ofthecolectomyonthe
plasmafattyacidpattern
UCpatients
Operated Non-operated Controls
pmolldl (n=15) (n=24) (n=107) p 16:0* 121-3(7 3) 1556(6-1) 128-9(3 4) 00013§11 16:It 6-6(10-10-3) 85(66-12-9) 8-4(8-1-9 5) 0 1631 18:0* 52-2(2 2) 60-1(2 8) 53-2(1-3) 0-0615 18:1* 90 1(6-6) 118 3(7-0) 110 6(3 2) 0031911 18:2n6* 147-5(8 6) 169-1(9 0) 137-6(3 9) 00037§ 18:3n3t 0 0(00-49) 2-1(0-05-9) 0 0(00-00) 0-0808 18:3n6t 0o0(00-1 9) 0-0(0-01-6) 00(00-00) 0-3176 20:2n6t 00(00-23) 05(00-2 1) 1-0(0-01-8) 07593 20:3n6* 12-7(1-4) 156(0 9) 13-3(0-5) 0-1082 20:4n6* 45-4(2 4) 52-0(2 6) 40-6(1-4) 0-0022§ 20:5n3t 0 0(00-23) 1-8(00-3 1) 07(00-1-7) 0 6853 22:6n3* 13-9(96-16.1) 16-7(11-7-19-7) 7-0(6-1-7-9) <0-00001tslI
*Mean(SEM). One wayANOVA+Scheffetest;tmedian(95%CI),Kruskall-Wallis test+Mann-Whitney U test;toperatedvcontrols;§non-operatedvcontrols;Iloperatedvnon-operated.
TABLE V Plasmafattyacids in inactive ulcerative colitis. Influenceofthecolectomyonthe
plasmafatty acid pattern UCpatients
Operated Non-operated Controls
% (n-15) (n=24) (n=107) p 16:0* 24-4(0-5) 257(0 3) 252(0-2) 0-2147 16:It 1-2(0-2-2-1) 1-4(0-9-2-1) 1-7(1-5-1-9) 0-0236t 18:0* 10-7(0 3) 9-9(0 2) 10-5(0 1) 0-2266 18:1* 18-1(0 7) 19-4(0-7) 21-7(0-3) 00002#§ 18:2n6* 29-9(1-0) 27-8(0 9) 27-2(0-4) 0-0918 18:3n3t 0-3(0-01-2) 0-3(0-01-0) 00(0 0-00) 0-1199 18:3n6t 00(00-04) 0-0(00-02) 00(00-00) 03530 20:2n6t 0 0(00-04) 0-1(00-003) 0-1(00-003) 0-3782 20:3n6t 2-6(20-3-3) 2-4(22-29) 25(23-2-8) 0-9194 20:4n6t 9-1(8-3-10-6) 8-6(79-99) 7-8(7-1-8-7) 0 0625 20:5n3t 00(00-09) 0-2(00-05) 0-2(00-003) 0-9017 22:6n3t 2-3(1-8-2.8) 2-6(1-9-3-5) 1-4(1-3-1-6) <0o00001ts UNID* 142-0(1-7) 141-7(1-7) 131-5(1-1) <0-OO001t§
*Mean(SEM). One wayANOVA+Scheffetest;tmedian(95% CI),Kruskall-Wallis test+Mann-Whitney U test;toperatedvcontrols; §non-operatedvcontrols.UNID=unsaturationindex.
UNID were also significantly high, both in
inactive ulcerative colitis and Crohn's disease, compared with controls (Table III).
Changes in saturated and monoenoic fatty acids depended ona diminished percentage of oleic acid(C18:1n9)and anincreased
concentra-tionofpalmitic acid(C16:0) inulcerativecolitis with respect to the control group and an
in-creasedpercentageofpalmitoleic acid(C16:1)in
Crohn's disease(Table III).
After adjusting for age as described in the statistical methods section, the significance of
thedifferencesseendidnotchange.
INFLUENCE OF COLECTOMY FORULCERATIVE
COLITIS ON PLASMA FATTY ACID PATTERN
(TablesIVandV)
Plasma docosahexaenoic acid (C22:6n3), either
as apercentage oranabsoluteconcentration,was
significantly higher both in operated and
non-operated ulcerative colitis patients when
com-paredwith controls. Inaddition,whenexpressed
as plasma concentration, C22:6n3 was signific-antly higher in non-operated than in operated
patients (Table IV). UNID was alsosignificantly
increased in both groups ofpatients compared with controls (Table V).
In the n6 series, the plasma concentration
of linoleic (C18:2n6) and arachidonic acid (C20:4n6) was only significantly increased in non-operated ulcerative colitis patients with
respecttothe controlgroup,
although
thesame trendwas seenfor theoperated
group.The increase of plasma
long
chain PUFAcontentin bothgroupsof
patients
wasassociatedwitha significantdecrease in the percentage of oleic acid(C18:1n9) in bothgroupsof
patients
and by the reduction in the percentage of
palmitoleic acid (C16:1n7) in
operated
patients
(TableV).
Afteradjustingfortheeffectofage,theresults didnotchangeexceptfor the oleic acid concen-tration,which showednodifferencesamongthe threegroups(p=0 151).
Discussion
Before discussing the results of this
study,
anaspectof the methodsused hastobe noted.Fatty
acid valuesin this paperareexpressedbothas a
percentage of the total
fatty
acids and as an absolute plasma concentration.Expressing
theresultsas plasmaconcentration
permitted
us toshowthatanychangeinagiven
fatty
acid isreal,
whereas its percentage mayonlyreflect compen-satorychangesbecauseofincreasesordecreases
inotherfattyacids. Theresults, however,should also begivenas apercentagedistribution. This
providescomplementaryinformation,asithelps
to ascertain whether changes in the
plasma
concentrations resultinvariationsin therelative
amounts of fatty acids - that is, in qualitative changesofplasmalipids.
Inapreviouspaper,9wereportedincreasedn3
anddecreasedn6PUFA values in activeIBD. It was then hypothesised that in active IBD a primary enhancement in PUFA synthesis co-existed with an increased utilisation related to the inflammatory phenomena. In this series of inactiveIBDpatients, plasman3PUFAarealso
increased.This, togetherwithhighvaluesofn6
PUFA and UNID, in the absence of factors associated with activedisease, further supports
this hypothesis. The fact that high n3 PUFA
values occur in both active and non-active
diseasecanbeexplainedbecause n3serieshas the
highestaffinity forPUFA biosynthetic enzyme systems.920
Theincrease in n6longchainPUFAis
partic-ularly noticeable in non-active,
non-operated
ulcerative colitis patients, their values
being
significantly higher than those in
healthy
controls. Also, significantly increased values of
the precursors of both n3 (C18:3n3) and n6
(C 18:2n6) series were seen in this group of
patients.Asahighintakeof essential
fatty acids,
particularly of a-linolenic acid (C18:3n3), was ruled out in all cases, this
finding
could beattributedto anegativefeedback effect2'-23upon
6-6
desaturaseactivity, caused by theexcessiveamountofproductsof both n3
(C22:6n3)
andn6 series (C20:4n6). From thesedata,
it could bespeculated that the hypothesis of an increased PUFAbiosynthesis is more active in ulcerative colitis than in Crohn's diseasepatients.
Differences in age distribution may account, atleast inpart, for thedifferencesseeninplasma fattyacid concentrations between operated and
non-operated patients and between ulcerative colitis patients and controls. After adjustingfor
Abnormal plasmapolyunsaturatedfatty acid pattern in non-active inflammatory bowel disease 1373
age as a covariate, however, the significance of the results did not change, except for oleic acid concentration, which showed no differences between operated and non-operated ulcerative
colitispatients.
Malnutritionmay be another important factor
thatexplains fatty aciddifferences among groups because PUFAdeficiency has been describedin
malnourished patients.2425 Malnutrition only *occurred, however, in four (10%) ulcerative colitisandthree(11%)Crohn'sdiseasepatients
in this series. Moreover, vitamin and trace elementdeficiencies, which have been reported in active IBD,2627 could be additional factors influencing PUFA biosynthesis. Unfortunately, dataon the micronutrient state in this series of
non-activepatients are not available.
Afurther argument in favour of the existence ofanincreased PUFAbiosynthesisinIBD is the persistence of increased values of docosahex-aenoic acid(C22:6n3) threeto six months after colectomy inpatientswithulcerative colitis. In
addition, this finding suggests that it may be becauseofaprimary metabolic defect. Although three to sixmonths seemsto beenough timeto reach a stable state after colectomy, the possi-bility that plasmaPUFApatterntooklongerto return to normal has to be ruled out in future studies.Thus, astudy comparing larger groups of patients with both proctocolectomy and colectomy plus ileal pouchpatients,atleasttwo
years after thecompletion of surgery, hastobe performedtoconfirmtheseresults.
On the other hand, the hypothesis of a
genetically determinedprimary defectinPUFA metabolism in IBD should be assessed by measuring PUFA profile in healthy relatives of IBDpatients.
These results further supportour reluctance
to use long chain n3 PUFA in treatment for
either acuteattacks ofIBD orfor maintenance
treatmentof the disease.
Supported bygrant88/2074 of theFISss,National Instituteof Health,Spain.
1 MeadJF. The non-eicosanoid functions oftheessentialfatty acids.JLipidRes 1984; 25: 1517-21.
2 Spector AA, Yorek MA. Membranelipidcomposition and cellularfunction.JLipid Res 1985; 26: 1015-35.
3 Kinsella JE, Lokesh B, Broughton S, Whelan J. Dietary polyunsaturated fatty acids and eicosanoids: Potential effectsonthe modulation ofinflammatoryand immunecells:
Anoverview.Nutrition1990;6:24-44.
4 Robinson DR. Alleviation ofautoimmune disease by dietary lipids containing omega-3fatty acids. Rheum Dis ClitNorth Am1991; 17: 213-22.
5 Falardeau P, Hamberg M, Samuelsson B. Metabolism of 8,11,
14-eicosatrienoic acid in human platelets. Biochim Biophys Acta1976; 441: 193-200.
6 Samuelsson B, Goldyne M, Granstrom E, Hamberg M, Hammarstrom S,Malmsten C. Prostaglandins and throm-boxanes. AnnRevBiochem1978; 47: 997-1029.
7 SamuelssonB.Leukotrienes: mediators of immediate hyper-sensitivity reactions andinflammation. Science 1983; 220: 568-75.
8 NeedlemanP,Raz A,Minkes MS, Ferrendelli JA, Sprecher H. Trieneprostaglandins: Prostacyclin and thromboxane biosynthesis and unique biological properties. Proc Natl Acad SciUSA1979;76: 944-8.
9Esteve-Comas M, Ramirez M,Fernandez-Baniares F, Abad-LacruzA, Gil A,Cabre E, et al. Plasma polyunsaturated fatty acid pattern in activeinflammatoryboweldisease. Gut
1992;33: 1365-9.
10 TrueloveSC, WittsLJ.Cortisone in ulcerative colitis. Final report on atherapeutic trial.BMJ1955; 2: 1041-6.
11 Van Hees PAM, Van Elteren PH, Van Lier HJJ, Van
Tongeren JHM. An index of inflammatory activity in patients with Crohn's disease. Gut1980; 21: 279-86. 12 Gassull MA, Cabre E, Vilar L, Alastrue A, Montserrat A.
Protein-energy malnutrition: Anintegral approach and a simple newclassification. Hum Nutr Clin Nutr 1984; 38C: 419-31.
13 Cabre E,Periago JL, Mingorance MD, Fernandez-BafiaresF, AbadA, Esteve M, et al. Factors relatedtotheplasmafatty acid profile in healthy subjects with special reference to antioxidant micronutrient status: a multivariate analysis. AmJ Clin Nutr1992; 55: 831-7.
14 Cabre E,Gonzalez-Huix F, Abad-Lacruz A, Gonzflez-Huix F,Gonzalez J, Esteve M, et al. Plasma fatty acid profile in
advanced cirrhosis: unsaturation deficit ofplasma lipid fractions.AmJGastroenterol1990; 85: 1597-604. 15 Galli C, White HB, Paoletti R. Brain lipid modifications
inducedby essentialfatty acid deficiencyingrowing male andfemalerats.J Neurochem 1970;17:347-55.
16 Norusis MJ. Advanced statistics SPSS/PC+ for the IBM PC/XT/AT.Chicago: SPSS Inc, 1986.
17 Diem K, LentnerC, eds. DocumentaGeigy. Scientific tables. 7thed.Basel: Geigy, 1970.
18 Zakim D. Metabolism ofglucose andfatty acids by the liver.
In: Zakim and Boyer, eds. Hepatology. Vol 1. 2nd ed.
Philadelphia:WBSaunders,1990:65-96.
19 Holman RT. Nutritional and metabolic interrelationships betweenfatty acids. Fed Proc 1964; 23:1062-7.
20 BrennerRR, Peluffo RO. Effect ofsaturatedand unsaturated
fattyacidsonthedesaturationinvitro ofpalmitic,stearic,
oleic, linoleic and linolenic acids.J Biol Chem1966; 241:
5213-9.
21 BivinsBA, Bell RM, Rapp RP, Griffen WO. Linoleic acid versus linolenic acid: What is essential?JPEN 1983;7: 473-8.
22 BrennerRR, PeluffoRO, Nervi AM,DeTomasME. Com-petitive effect ofa-andT-linolenyl-CoAand
arachidonyl-CoA in linoleyl-CoA desaturation to T-linolenyl-CoA.
BiochimBiophys Acta1%9;176:420-2.
23 Brenner RR, Peluffo RO. Inhibitory effect of
docosa-4,7,10,13,16,19-hexaenoic acid upon the oxidative desat-uration of linoleic into T-linolenic acid and ofa-linolenic acid
into octadeca-6,9,12,15-tetraenoic acid. Biochim Biophys
Acta 1967;137:184-6.
24 HolmanRT, JohnsonSB,Mercuri0,ItarteHJ,Rodrigo MA,
DeTomas ME. Essential fatty acid deficiency in
mal-nourished children.AmJClin Nutr1981;34:1534-9.
25 Wolff JA, Margolis F, Bujdoso-Wolff K, Matusick E, MacLeanWC. Plasma andredbloodcellfattyacid
com-position in children with protein-calorie malnutrition. PediatrRes1984; 18:162-7.
26 Fernandez-BaniaresF, Abad-L A, XiolX,Gine JJ,DolzC,
Cabre E,etal.Vitaminstatusinpatientswithinflammatory bowel disease.AmJ Gastroenterol 1989;84: 744-8. 27 Fernandez-Baniares F, Mingorance MD, Esteve-Comas M,
Cabre E,LachicaM, Abade-LacruzA,etal. Serumzinc,
copper and selenium levelsininflammatoryboweldisease.
doi: 10.1136/gut.34.10.1370
1993 34: 1370-1373
Gut
M Esteve-Comas, M C Núñez, F Fernández-Bañares, et al.
disease.
pattern in non-active inflammatory bowel
Abnormal plasma polyunsaturated fatty acid
http://gut.bmj.com/content/34/10/1370
Updated information and services can be found at:
These include: References
http://gut.bmj.com/content/34/10/1370#related-urls
Article cited in:
service Email alerting
box at the top right corner of the online article.
Receive free email alerts when new articles cite this article. Sign up in the
Collections Topic (941 articles) Ulcerative colitis (792 articles) Crohn's disease
Articles on similar topics can be found in the following collections
Notes
http://group.bmj.com/group/rights-licensing/permissions
To request permissions go to:
http://journals.bmj.com/cgi/reprintform
To order reprints go to:
http://group.bmj.com/subscribe/