Appendice
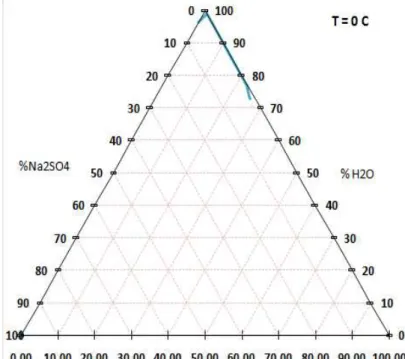
Tabelle e dati di solubilità.Fig 12: Solubilita` a 0 ° C
%wt H20 %wt Nacl % wt Na2SO4 fasi solide 72,89 25,79 1,32 CL 2+S 10+ CL 73,31 25,30 1,39 CL 2+S 10+ CL 77,08 22,8 0,12 ICE+CL 3+S 10 76,26 23,43 0,31 ICE+CL 2+S 10 99,08 22.09 0 ICE+CL 2 99,02 23.18 0,01 ICE+CL 2 96,21 0 3,79 ICE+ S 10
Fig 13: Solubilita` a 10 ° C
%wt H20 %wt Nacl % wt Na2SO4 fasi solide
91,23 0 8,77 NA2SO4 10 H20 90,33 3,87 5,8 NA2SO4 10 H20 87,45 8,39 4,16 NA2SO4 10 H20 87,15 8,78 4,07 NA2SO4 10 H20 83,58 13,08 3,34 NA2SO4 10 H20 79,54 17,2 3,26 NA2SO4 10 H20 79,49 17,35 3,16 NA2SO4 10 H20 75,59 21,27 3,14 NA2SO4 10 H20 74,38 22,39 3,23 NA2SO4 10 H20 73,07 23,58 3,35 NA2SO4 10 H20 72,02 24,45 3,53 NA2SO4 10 H20 NaCl 73,68 26,32 0 NA2SO4 10 H20 NaCl
Fig 14: Solubilita` a 15 ° C
%wt H20 %wt Nacl % wt Na2SO4 fasi solide
84,50 0 15,5 S 10 86,72 5,42 7,86 S 10 82,62 11,51 5,87 S 10 78,80 15,97 5,23 S 10 73,71 21,03 5,26 S 10 70,97 23,39 5,64 S 10+ Cl 71,39 23,20 5,41 S 10+ Cl 72,53 25,21 2,26 Cl 73,65 26,35 0,00 Cl
Fig 15: Solubilita` a 25 ° C
%wt H20 %wt Nacl % wt Na2SO4 fasi solide
77,79 0 22,209 NA2SO4 10 H20 77,19 2,278 20,532 NA2SO4 10 H20 76,86 4,450 18,691 NA2SO4 10 H20 76,16 7,539 16,304 NA2SO4 10 H20 75,16 10,093 14,746 NA2SO4 10 H20 73,81 11,698 14,495 NA2SO4 10 H20 71,50 13,677 14,821 NA2SO4 10H20 NA2SO4 71,68 16,645 11,670 NA2SO4 10H20 NA2SO4 71,86 19,071 9,068 NA2SO4 10H20 NA2SO4 70,90 22,022 7,076 NA2SO4 10H20 NA2SO4 70,25 22,769 6,976 NA2SO4 NACL 70,01 23,102 6,888 NA2SO4 NACL 71,19 24,069 4,741 NA2SO4 NACL 72,47 25,076 2,449 NA2SO4 NACL 73,64 26,362 0 NACL
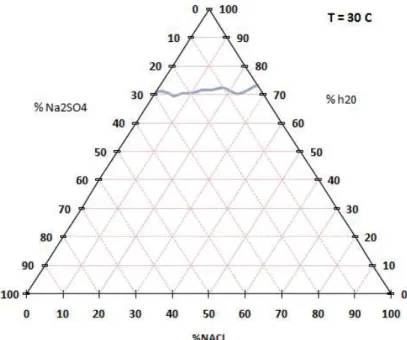
Fig 16: Solubilita` a 30 ° C
%wt H20 %wt Nacl % wt Na2SO4 fasi solide
70,90 0 29,10 S 10 71,07 1,74 27,19 S10 70,65 3,35 26 S 10 70,37 3,95 25,68 S10 69,51 5,5 24,99 S10 69,32 5,68 25 S 10 + S 70,30 7,46 22,24 S 10 + S 70,31 8,69 21 S 10 + S 71,08 11,13 17,79 S 10 + S 71,60 12,2 16,2 S 71,56 13,2 15,24 S 71,57 14,79 13,64 S 72,30 18 9,7 S 70,32 23 6,68 S + Cl 73,49 26,51 0 Cl
Fig 16: Solubilita` a 50 ° C
%wt H20 %wt Nacl % wt Na2SO4 fasi solide
68,40 0 31,6 71,35 7,85 20,8 S 72,60 16,1 11,3 S 70,36 24,1 5,54 S + cl 71,94 25,5 2,56 CL 73,14 26,86 0
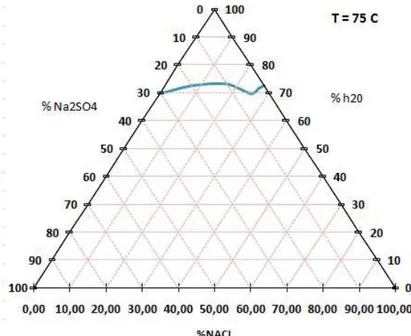
Fig 16: Solubilita` a 75 ° C
%wt H20 %wt Nacl % wt Na2SO4 fasi solide
69,50 0 30,5 72,47 7,76 19,77 S 73,20 16,5 10,3 S 69,75 25,3 4,95 S + cl 71,46 26,4 2,14 Cl 72,60 27,4 0
Fig 17: Solubilita` a 100 ° C
%wt H20 %wt Nacl % wt Na2SO4 fasi solide
70,40 0 29,6 S 73,73 7,67 18,6 S 72,85 18,4 8,75 S 69,59 25,9 4,51 S + cl 70,96 27,2 1,84 Cl 71,85 28,15 0
Bibliografia
• Mullin J.W. Crystallization 4th Edition Butterworth Heinemann 2001.
• Donald E. Garret Sodium Sulfate: handbook of deposits, processing, properties and use. Academic Press 2001.
• Miller, Donald G.; Ting, Alexander W.; Rard, Joseph A.; Eppstein, Lee B. Ternary
diffusion coefficients of the brine system sodium chloride (0.5 sodium sulfate (0.5 M)-water and sodium chloride (0.489 M)-magnesium chloride (0.051 M)-M)-water (seaM)-water composition) at 25 C. Geochimica et Cosmochimica Acta (1986), 50(11), 2397-403.
CODEN: GCACAK ISSN: 0016-7037. Journal written in English. CAN 106:22920 AN 1987:22920 CAPLUS (Copyright (C) 2010 ACS on SciFinder (R))
• Suljkanovic, Midhat; Ahmetovic, Elvis; Kravanja, Zdravko. The process simulator of
alternative process structures of fractional crystallization from the NaCl-Na2SO4-H2O system. Faculty of Technology, University of Tuzla, Tuzla, Bosnia/Herzegovina.
Slovenski Kemijski Dnevi, Maribor, Slovenia, Sept. 25-26, 2003 (2003), 715-723. Publisher: Univerza v Mariboru, Fakulteta za Kemijo in Kemijsko Tehnologijo, Maribor, Slovenia CODEN: 69FCOE Conference; Computer Optical Disk written in Slovenian. CAN 140:201612 AN 2004:199322 CAPLUS (Copyright (C) 2010 ACS on
SciFinder (R))
• Wang, Che-li; Qiu, Zhao-rong; Cheng, Min-lian; Ye, Qing; He, Song-bo. Separation of
sodium sulfate and sodium chloride from brines by solventing-out process. Department
of Chemical Engineering, Jiangsu Polytechnic University, Changzhou, Peop. Rep. China. Gaoxiao Huaxue Gongcheng Xuebao (2003), 17(6), 711-714. Publisher: Zhejiang Daxue, CODEN: GHGXEG ISSN: 1003-9015. Journal written in Chinese. CAN 140:61742 AN 2004:26705 CAPLUS (Copyright (C) 2010 ACS on SciFinder (R))
• Abdullaev, K. M.; Agamaliev, M. M.; Sultanova, F. M.; Krikun, M. M.; Dadasheva, G. I.
Technology of utilization of waste brines from water desalination plants. Azerb. Inst.
Neftekhim., Baku, Azerbaijan. Khimiya i Tekhnologiya Vody (1992), 14(11), 863-9. CODEN: KTVODL ISSN: 0204-3556. Journal written in Russian. CAN 118:105757 AN 1993:105757 CAPLUS (Copyright (C) 2010 ACS on SciFinder (R))
R. Grace and Co., Precipitation of sulfate salts from saline solutions. Clarksville, MD, USA. Avail. GPO. U. S. Dep. Interior, Office Saline Water Res. Develop. Progr. Rep. (1968), (5. pp.), 59 pp. CODEN: XISWAP Report written in English. CAN
69:61447 AN 1968:461447 CAPLUS (Copyright (C) 2010 ACS on SciFinder (R))
• Lebedev, N. V.; Bannikova, A. A. The system of sodium sulfate-water. Sb. Tr. Gos. Nauchn.-Issled. Inst. Gidrolizn. i Sul'fitno-Spirt. Prom. (1962), 10(154-65), 230. From: Ref. Zh., Khim. 1963, Abstr. No. 16P17. Journal language unavailable. CAN 60:72165 AN 1964:72165 CAPLUS (Copyright (C) 2010 ACS on SciFinder (R))
• Rafols, Jose. Sodium sulfate recovery. Ing. Quim. (Mex.) (1963), 8(81), 26-9. Journal language unavailable. CAN 60:50931 AN 1964:50931 CAPLUS (Copyright (C) 2010 ACS on SciFinder (R))
• Ravich, M. I.; Borovaya, F. E. Phase equilibriums in the quaternary system sodium
sulfate-sodium chloride-sodium hydroxide-water at 350 and 400 Zhurnal
Neorganicheskoi Khimii (1959), 4 2100-15. CODEN: ZNOKAQ ISSN: 0044-457X. Journal language unavailable. CAN 54:60413 AN 1960:60413 CAPLUS
(Copyright (C) 2010 ACS on SciFinder (R))
• Ravich, M. I.; Borovaya, F. E.; Ketkovich, V. Ya. Phase equilibria at high temperatures
in the system NaCl-Na2SO4-H2O. Doklady Akademii Nauk SSSR (1951), 77 617-20.
CODEN: DANKAS ISSN: 0002-3264. Journal language unavailable. CAN 48:9846 AN 1954:9846 CAPLUS (Copyright (C) 2010 ACS on SciFinder (R))