CD200 and Chronic
Lymphocytic Leukemia: Biological
and Clinical Relevance
Giovanni D
’Arena
1*
, Vincenzo De Feo
2, Giuseppe Pietrantuono
3, Elisa Seneca
3,
Giovanna Mansueto
3, Oreste Villani
3, Francesco La Rocca
4, Fiorella D
’Auria
5,
Teodora Statuto
5, Luciana Valvano
5, Francesca Arruga
6, Silvia Deaglio
6,
Dimitar G. Efremov
7, Alessandro Sgambato
8and Luca Laurenti
91Hematology,“S. Luca” Hospital, ASL Salerno, Vallo della Lucania, Italy,2Department of Pharmaceutical and Biomedical Sciences, University of Salerno, Salerno, Italy,3Hematology and Stem Cell Transplantation Unit, IRCCS Centro di Riferimento Oncologico della Basilicata, Rionero in Vulture, Italy,4Laboratory of Preclinical and Translational Diagnostics, IRCCS Centro di Riferimento Oncologico della Basilicata, Rionero in Vulture, Italy,5Laboratory of Clinical Research and Advanced Diagnostics, IRCCS Centro di Riferimento Oncologico della Basilicata, Rionero in Vulture, Italy,6Cancer Immunogenetics Unit, Department of Medical Sciences, Molecular Biotechnology Center, University of Turin, Turin, Italy,7Molecular Hematology, International Centre for Genetic Engineering and Biotechnology, Trieste, Italy,8Scientific Direction, IRCCS Centro di Riferimento Oncologico della Basilicata, Rionero in Vulture, Italy,9Hematology Institute, IRCCS Fondazione Policlinico Universitario A. Gemelli, Rome, Italy
CD200, a transmembrane type Ia glycoprotein belonging to the immunoglobulin protein
superfamily, is broadly expressed on a wide variety of cell types, such as B lymphocytes, a
subset of T lymphocytes, dendritic cells, endothelial and neuronal cells. It delivers
immunosuppressive signals through its receptor CD200R, which is expressed on
monocytes/myeloid cells and T lymphocytes. Moreover, interaction of CD200 with
CD200R has also been reported to play a role in the regulation of tumor immunity.
Overexpression of CD200 has been reported in chronic lymphocytic leukemia (CLL) and
hairy cell leukemia but not in mantle cell lymphoma, thus helping to better discriminate
between these different B cell malignancies with different prognosis. In this review, we focus
on the role of CD200 expression in the differential diagnosis of mature B-cell neoplasms and
on the prognostic signi
ficance of CD200 expression in CLL, where conflicting results have
been published so far. Of interest, increasing evidences indicate that anti-CD200 treatment
might be therapeutically bene
ficial for treating CD200-expressing malignancies, such as CLL.
Keywords: CD200, chronic lymphocytic leukemia, prognosis, diagnosis,flow cytometry
INTRODUCTION
Mature B-cell leukemias are heterogeneous in clinical and biological features. Despite the large body
of studies published, dif
ficulties to get a firm diagnosis still exist in some cases due to the lack of
disease-speci
fic markers and overlapping immunophenotypes. CD200 has recently emerged as a
useful tool to better discriminate among several chronic leukemias. In addition to the usefulness of
this marker in the diagnostic setting, it also has a prognostic role and may represent a potential
therapeutic target in chronic lymphocytic leukemia (CLL).
Frontiers in Oncology | www.frontiersin.org 1 November 2020 | Volume 10 | Article 584427
Edited by: Je´ roˆme Paggetti, Luxembourg Institute of Health, Luxembourg Reviewed by: Jorge Castillo, Dana–Farber Cancer Institute, United States Michael Mian, Ospedale di Bolzano, Italy *Correspondence: Giovanni D’Arena [email protected]
Specialty section: This article was submitted to Hematologic Malignancies, a section of the journal Frontiers in Oncology Received: 17 July 2020 Accepted: 27 October 2020 Published: 26 November 2020 Citation: D’Arena G, De Feo V, Pietrantuono G, Seneca E, Mansueto G, Villani O, La Rocca F, D’Auria F, Statuto T, Valvano L, Arruga F, Deaglio S, Efremov DG, Sgambato A and Laurenti L (2020) CD200 and Chronic Lymphocytic Leukemia: Biological and Clinical Relevance. Front. Oncol. 10:584427. doi: 10.3389/fonc.2020.584427
REVIEW published: 26 November 2020 doi: 10.3389/fonc.2020.584427
In this review, we provide a summary of published data on the
biological and clinical relevance of CD200 in CLL, identified
through a literature search of the MEDLINE, Google Scholar,
and Scopus databases, aiming at providing an update of the
published literature on this topic. The search comprised the
terms
“CD200”, “chronic lymphocytic leukemia”, and “chronic B
cell leukemias
” without a date restriction. All articles and
Meeting Abstracts we found were evaluated and included in
this review.
CD200 ANTIGEN AND CD200 RECEPTOR
The surface membrane glycoprotein CD200, formerly termed
OX-2, is encoded by the 29,744 bp long CD200 (OX-2) gene
located on the long arm of chromosome 3 (3q13.2) (
1
).Three
transcript variants of CD200 are known: variant 1, 2,226 bp long,
containing 7 exons and encoding the 269 amino acids long
isoform a; variant 2, 2,301 bp long, containing 7 exons and
encoding the 294 amino acids long isoform b; and variant 3,
2,085 bp long, missing an exon and encoding the 153 amino
acids long isoform c (
2
).
The CD200 glycoprotein is a single-pass, type I, highly
conserved membrane protein, belonging to the immunoglobulin
superfamily, spanning the membrane once, with the N-terminus
on the extracellular side of the membrane (
2
). It is composed of two
extracellular (one variable and one constant) immunoglobulin-like
domains, a single transmembrane region, and a cytoplasmic tail
(
Figure 1
) (
3
).
The receptor for CD200 (CD200R) also has two
immunoglobulin-like domain. The phylogenetic analysis
demonstrated that the receptor is closely related to CD200 and
probably evolved by a gene duplication (
2
). As a matter of the fact,
the genes for human CD200 and CD200R are closely linked on
chromosome 3 (
4
). However, the CD200R has a longer
cytoplasmic tail with signaling motifs different from CD200 (
5
).
CD200 is normally expressed on a variety of cell types,
including thymocytes, B lymphocytes, a subset of T
lymphocytes, neurons, endothelial cells, some dendritic cells,
kidney glomeruli, and syncytiothrophoblasts (
6
). The expression
of CD200R is more restricted and includes myeloid leucocytes,
such as macrophages, dendritic cells, and mast-cells, as well as B
lymphocytes and a subset of T lymphocytes (
7
,
8
).
FUNCTION OF CD200 AND ITS ROLE
IN CANCER
CD200, by means of the interaction with its receptor, induces the
suppression of T-cell mediated responses, limiting in
flammation
in a wide range of in
flammatory diseases (
Table 1
). The
inhibition of macrophage function, induction of regulatory
T-helper cell type (Th1) to Th2 cytokine profile switch, suppression
of natural killer cell function, and inhibition of tumor-specific
T-cell immunity have been all experimentally demonstrated (
9
–
14
,
15
–
18
). Consistent with its immunosuppressive role,
CD200-deficient mice are susceptible to tissue-specific autoimmunity
(
15
). Gorczynski and co-workers demonstrated that the
interaction of CD200 with CD200R is able to decrease the
production of Th1-like cytokines, such as interleukin (IL)-2 and
interferon (IF)-g, and increase the release of Th2-like cytokines,
such as IL-10 and IL-4 (
16
). In addition, the same group reported
that the CD200/CD200R interaction induces the in vitro
differentiation of T lymphocytes toward CD4
+CD25
+Foxp3
+regulatory T-cells (Tregs) (
16
).
Moreaux et al demonstrated that CD200 mRNA is
overexpressed on cells of several types of cancers compared to
their normal counterparts, including chronic lymphocytic
leukemia (CLL) (
19
). Kretz-Rommel et al elaborated a tumor
model on the basis of the previous demonstration that CD200 is
up-regulated in CLL and that the up-regulation in multiple
myeloma (MM) and acute myeloid leukemia (AML) correlates
with adverse prognosis (
20
–
22
). These authors
firstly
demonstrated that human peripheral blood mononuclear cells
(hPBMCs) and Namalwa tumor cells (Burkitt
’s lymphoma cell
line lacking CD200 expression) simultaneously injected in
FIGURE 1 | Schematic representation of CD200. The CD200 glycoprotein has two extracellular like Immunoglobulin domains formed by disulfide bonds, one variable (V) and one constant (C), a single transmembrane region, and a cytoplasmic tail.
TABLE 1 | CD200:CD200R interaction and negative control of immunity.
KEY FACTS
Reduced Th1 cytokine (IL-2, IFNg) production (9) Increased IL-10 and IL-4 production (9) Induction of Tregs (10)
Inhibition of mast cell degranulation (11,12) Downregulation of basophilic function (13) Suppression of natural killer cell function (14)
CD200R, receptor of CD200 antigen; Th, T helper; IL, interleukin; IFN, interferon; Tregs, regulatory T-cells.
D’Arena et al. CD200 and CLL
NOD/SCID mice show reduced tumor growth with respect to
that observed in mice in the absence of hPBMCs (
23
,
24
). When
the Namalwa tumor cells were engineered to express human
CD200 on their surface and simultaneously injected with
hPBMCs, CD200 expressed on tumor cells prevented hPBMCs
from eradicating the cancer cells. This tumor model clearly
demonstrated the immunosuppressive activity of CD200.
Moreover, treatment with anti-CD200 monoclonal antibodies
(Abs) was also highly effective in this model and inhibited the
growth of Namalwa CD200 tumor cells in NOD/SCID hu-mice
by >90%. The same study showed that CD200 is a marker of
activated T cells and that the use of IgG1 anti-CD200 (a constant
region variant that can mediate ADCC) resulted in ef
ficient
target cell killing of these lymphocytes by ADCC. Taken
together, these data have relevant implications for the
immunotherapy of cancer patients with anti-CD200.
RELEVANCE OF CD200 IN DIAGNOSIS OF
CHRONIC LYMPHOPROLIFERATIVE
DISORDERS
The neoplasms of mature B cells are heterogeneous diseases
currently included in the
“mature B-cell lymphoid neoplasms” of
the WHO classification (
25
,
26
). Immunophenotypic profile,
cytogenetics, and molecular biology must be all taken into
account as a multidisciplinary integrated approach to
differentiate the disease entities belonging to this category.
However, difficulties in defining some cases still exist.
Diagnostic accuracy is crucial in diagnosing neoplasms that
require different speci
fic treatments.
Flow cytometric characterization of chronic lymphoproliferative
disorders represents a cornerstone in the diagnostic approach to
neoplasms of mature lymphocytes (
27
). In the early 90s, a British
group in London proposed a scoring system (Matutes score),
which was based on analysis of 5 membrane markers: CD5,
CD22, CD23, FMC7, and surface immunoglobulin (SmIg) (
28
).
A score of 1 was assigned for each of the following
immunophenotypic features: CD5 positive, CD22 weak or
negative, CD23 positive, FMC7 negative, SmIg weak. The total
score, according to this scoring system, is usually 4 or 5 for
typical CLL cases, and 3 or less for other mature B-cell lymphoid
neoplasms (
28
). A few years later, the same group improved the
diagnostic accuracy of the score (from 91.8% to 96.8%) simply
replacing CD22 with CD79b (
29
).
The Matutes score is used worldwide as a diagnostic tool.
However, some cases of chronic lymphoid neoplasms are still
misdiagnosed, including some cases belonging to a more
aggressive entity, such as mantle cell lymphoma (MCL). In this
context, CD200 has been shown to have differential expression
in B-cell neoplasms and to well discriminate CLL from MCL and
hairy cell leukemia (HCL) and its variant form (v-HCL)
(
Figure 2
) (
30
–
32
).
The reports on the relevance of CD200 expression to
differentiate B cell chronic lymphoid neoplasms that have been
published so far are listed in
Table 2
. Firstly, Palumbo et al in
2009 demonstrated that CLL can be differentiated from MCL
according to the expression of CD200 (all patients with CLL
expressed CD200, whereas all patients with MCL were negative)
(
31
). At the same time Brunetti et al, analyzing only patients with
HCL, showed that all patients with typical HCL expressed
CD200 (
30
). Shortly after, Dorfman & Shahsafaei studied by
means of
flow cytometry bone marrow and lymph node aspirates
from patients with different mature B cell lymphoid neoplasms,
showing that CLL and HCL cases were all CD200 positive,
prolymphocytic leukemia (PLL) were positive in 80% of cases,
while MCL, splenic marginal zone lymphoma (SMZL), and
follicular lymphoma (FL) were all negative (
32
). Since then,
several other researchers analyzed samples from patients with
mature B-cell lymphoid neoplasms (
33
–
63
). Overall, the
published data con
firmed the positivity in all patients with
FIGURE 2 | CD200 expression in CLL and MCL. Dot plots showing a case of CLL in which both surface CD200 and CD23 were expressed (left panel) and a case of MCL in which both antigens were found negative (right panel). B-cells only have been gated for the analysis.
D’Arena et al. CD200 and CLL
TABLE 2 | CD200 expression and differential diagnosis in chronic B-cell leukemias: published data at a glance.
Reference No.
Patients
Samples evaluated CLL MCL HCL HCL-v MZL FL LPL Other B-cell neoplasms
Palumbo et al. (31) 91 PB 79/79 (100%) 0/14 (0%) – – – – – – Brunetti et al. (30) 10 PB; BM – – 10/10 (100%) – – – – – Dorfman and Shahsafaei (32) 73 BM, LN# 21/21 (100%) 0/10 (0%) 12/12 (100%) – 0/10 (0%) 0/16 (0%) 8/10 (80%)
MALT: 0/4 (0%); B lymphoblastic leukemia/ lymphoma: 10/10 (100%); DLBCL: 0/12 (0%); mediastinal large B-cell lymphoma: 8/ 8 (100%); BL: 0/8 (0%); MM: 10/13 (77%); HL: 12/13 (92%); nodular lymphocyte predominant HL: 0/14 (0%)
Bhatnagar et al. (33) 100 PB; BM 78/78 (100%) 0/7 (0%) – – – – – –
El Desoukey et al. (34) 49 PB 31/31 (100%) 0/4 (0%) 2/2 (100%) – 0/4 (0%) 0/8 (0%) – –
Cherian et al. (35) 66 Not reported – – – – – – – Not reported data as percentage
expression of CD200 in 51 cases of CLL/ SLL and 15 cases of MCL. Most cases studied can be separated using a cut off of 1.10 for the CD200 MFI ratio
Kern et al. (36) 100 PB, BM 58/59
(98,3%)
2/14 (14,3%) – – – – – CLL/PL cases: 26/27 (96.3%)
Alapat et al. (37) 107 PB; BM; LN; body fluids
19/19 (100%) 0/4 (0%) – – – – 3/7
(43%)
B-ALL: 19/20 (95%); T-ALL: 0/5 (0%); MM: 37/52 (71%)
Pillai et al. (38) 180 BM, PB, LN; body fluids and other tissues
– – 23/23 (100%) 1/1 0(10%) – – – Not specified the number of positive/
negative cases but only reported MFI (with range) for each disease category. CLL/SLL showed CD200 with MFI of 5,965 compared with MCL and FL which had MFIs of 397 and 521, respectively. Dasgupta et al. (39) 56 Not reported 28/28 (100%) 2/10
(20 %)
3/3 (100%) – – – – 4/8 (50%) not otherwise specified
El-Sewefy et al. (40) 40 Not reported 30/30 (100%) 1/10 (10%) – – – – –
Sandes et al. (41) 159 PB; BM; LN; CSF 56/56 (100%)** 0/14 (0%) 13/13 (100%) – 6/6 (100%) 8/11 (73%) 2/4 (50%) PL: 2/7 (28%) SRPBL: 1/1 (100%) CD5-CD10- unclassifiable NHL: 21/31 (68%) CD5+ NHL*** 9/23 (39%) Challagundla et al. (42) 364 PB; BM; FNA; LN 119/119 (100%) 58/61
(95%)* 7/7 (100%) – 9/26 (35%)° °° 3/3 (100%)°° Karban et al. (43) 200 PB 200/20 (100%) 4/46 (8,7%) – – – – – – (Continued) D’Arena et al. CD200 and CLL Frontiers in Oncology | www.frontiersin.org November 2020 | Volume 10 | Article 584427 4
TABLE 2 | Continued
Reference No.
Patients
Samples evaluated CLL MCL HCL HCL-v MZL FL LPL Other B-cell neoplasms
Sorigue et al. (44) 248 PB; BM; LN; CE 106/106 (100%) – – – – – – MBL-CLL like: 106/106 (100%); DLBCL: 7/
32 (22%) of which 4/11 (36%) ABC; 3/20 (15%) GCB; 0/1 (0%) cell of origin not determined; 1/4 (25%) BL. Lesesve et al. (45) 124 PB; BM 57/69 (83%) 0/10 (0%) 2/4 (50%) – 1/16 (6%) 0/7 (0%) – MBL-CLL like: 8(13(61%); MBL-non-CLL like 1/3(33%); PL: 0/2 (0%) Fan et al. (46) 374 PB; BM 268/271 (99 %)
1/31 (35%) 3/3 (100%) – 75% strong – – PL: 5/7 (%) weakly, 2/7 (%) strong Naseem et al. (47) 77 Not reported 54/54 (100%) 1/6 (16%) 5/5 (100%) – – 1/2 (50%) – Other CLPD did not express CD200
Rahman et al. (48) 3 Not reported – – – 3/3 (100%) – – – –
Rahman et al. (49) 160 PB; BM; FNA, ascitesfluid 98/98 (100%) 0/24 (0%) 6/6 (100%) 0/1 (0%) 3/6 (50%) 2/4 (50%) 2/4 (50%) DLBCL: 3/5 (60%); BL: 0/1 (0%); PBL: 0/1 (0%); CD5-CD10- undefined lymphomas: 4/10 (40%)
Ting et al. (50) 97 PB; BM; LN; pleural fluid
56/56 (100%)§
0/6 (0%) 2/2 (100%) – – – – 40 pts of which 22 diagnosed with
lymphomas with subtype (not reported in the paper), and 18 unclassified (of which 10 diagnosed with lymphoma without subtype, and 8 non-CLL MBL) Arlindo et al. (51) 124 PB; BM 61.1 (41.4/89.2) 3.5 (2.1/4.1) 220.3 (163.1/297.5) 36.1 (22.1/50.1) 8.3 (4.5/13.2) 2.6 (1.8/11.4) Atypical CLL: 113.7 (70.4/122.2) Mason et al. (52) 79 PB;BM;LN – – 34/34 (100%) 0/3 (0%) 1/22 (5%) – 13/20 (65%) Starostka et al. (53) 188 PB; BM; LN; pleural
fluid 158/161 (98.3%) 7.9% – – 63.6% – – Miao et al. (54) 653 PB; BM 355/365 (97%) 12/41 (29%) 1/1 (100%) 2/2 (66.7%) 8/20 (40%) 13/17 (76.5%) 30/35 (85.7%) MALToma: 4/4 (100%); CD5+ and CD5-unclassified B-cell chronic
lymphoproliferative disorders: 119/153 (78%) Poongodi et al. (55) 77 PB; BM 54/54 (100%) 1/6 (16,7%) 5/5 (100%) – 2/2 (100%) 1/2 (50%) – DLBCL: 1/3 (33.3%); unclassifiable lymphoma: 2/3 (66.7%); SLL: 1/1 (100%) Favre et al. (56) 96 PB 84/84 (100%) 1/30 (3%) 7/7 (100%) – 13/14 (93%) – – SRPBL: 6/15 (40%) Falay et al. (57) 339 PB; BM 295/306ç (95.8%) 2/33 (6.1% dimly) – – – – – D’Arena et al. (58) 427 PB; BM 312/322 (97%) 4/21 (19%) 15/15 (100%) – 27/53 (51%) 3/12 (25%) 0/4 (0%)
Debord et al. (59) 135 PB, BM – 3/63 (5%) – – – – – 68/72 (94%) low grade B-cell lymphoma
not otherwise specified
Mora et al. (60) 120 PB 64/64 (100%) 1/5 (20%) – – 13/19 (68.4%) 2/2 (100%) – MBL: 14/14 (100%); SLL: 3/3 (100%); other B-CLPD (not otherwise specified): 9/ 13 (69.2%)^
Myles et al. (61) 307 PB; BM; LNH, other bodyfluid or tissue
231/241 (96%) 62/66 (94%) – – – – – (Continued) D’Arena et al. CD200 and CLL Frontiers in Oncology | www.frontiersin.org November 2020 | Volume 10 | Article 584427 5
CLL and HCL and the negativity in almost all patients with MCL
(
Table 2
).
Focusing on the assessment of
fluorescence intensity of
CD200, Lesesve et al used the ratio of mean
fluorescence
intensity (MFI) of CD160/CD200 on leukemic cells/controls
(
45
). CD160 is a glycosylphosphatidylinositol-anchored cell
surface molecule belonging to the immunoglobulin superfamily
that was found to be expressed in patients with CLL (
64
,
65
).
Only 60% of patients showed surface positivity for CD160. Both
markers were positive in 55% of CLL but only in 2% of other
B-cell neoplasms, and absence of both markers occurred in 12% of
CLL and in 86% of other B-cell neoplasms.
Taken together, these data showed that CD200 has a high
sensitivity for CLL diagnosis, being expressed in most of the CLL
cases. However, it is expressed also in other B-cell malignancies,
including MZL, HCL, and even some cases with indolent,
non-nodal entity MCL, thus showing low speci
ficity.
Some authors tried to implement the diagnostic ability of
monoclonal antibodies-based scores using CD200 (
58
,
66
–
68
).
Recently, Köhnke et al proposed the so-called
“CLLflow score”
including CD200 in the Matutes score and showing an
improvement in the specificity for the diagnosis of CLL (
66
).
Brie
fly, the score is calculated by adding the percentage of
CD200+ and CD23+/CD5+ B cells and then subtracting the
percentages of CD79b+ as well as FMC7+ B cells. A score >0 is
consistent with the diagnosis of CLL while a score
≤ 0 with the
diagnosis of a non-CLL disease entity (
67
–
69
). In our hands a
simpli
fied score system for the diagnosis of CLL, in which only 4
markers are used (CD5, CD23, CD200, and SmIg), showed a
higher sensitivity and specificity with respect to the Matutes
score (
58
).
Interestingly, very recently Sorigue et al, using three
immunophenotype-based diagnostic approaches (Matutes
score, D’Arena score and CD43 expression) analyzed 597
patients with a chronic lymphoproliferative disorder (CLPD)
and found that patients with concurring CLL-like or non-CLL
like results according to the three diagnostic strategies were
diagnosed with CLL (n = 441) and non-CLL (n =99),
respectively (
Figure 3
) (
69
,
70
).
‘Discordant’ patients (n = 57)
were further re-evaluated taking into account individual
cytometric markers and cytogenetics data and only 16 patients
(2.7%) were not assigned to a reasonable diagnosis (
Figure 3
).
The latter cases were considered
“borderline lymphoproliferative
disorders
”, a loosely-defined concept that would include any
chronic lymphoid disorder in which the diagnosis of CLL cannot
be either made or ruled out.
In another recent study, Sorigue et al conducted a systematic
review of the use of CD200 in the differential diagnosis of CLPDs
(
71
). They evaluated the positive predictive value of CD200 on
the prevalence of the disorders evaluated for the differential
diagnosis. Twenty-seven publications were included in this
systematic review (accounting for 5,764 patients). The median
positivity rate and percentage of CD200 positive cells in patients
with CLL was 100% and 95%, respectively, whereas it was 4% and
8% in MCL, and 56% and 62% in other CLPDs. The authors
concluded that CD200 is a suboptimal marker in discriminating
TABLE 2 | Continued Reference No. Patients Samples evaluated CLL MCL HCL HCL-v MZL FL LPL Other B-cell neoplasms Soong et al. ( 62 ) 189 PB; BM 121/121 (100%) 1/10 (1%) –– – – – 54/68 of B-chronic lymphoid leukemias, including MCL cases El-Neanaey et al. ( 63 ) 5 0 P B 30/30 (100%) – 5/5 (100%) –– – – 14/25 (56%) B-NHL not subclassi fied CLL, chronic lymphocytic leukemia; MCL, mantle cell lymphoma; HCL, hairy cell leukemia; HCL-v, hairy cell leukemia variant; MZL, marginal zone lymp homa; FL, follicular lymphoma; LP, lymphoplasmacytic lymphoma; PL, prolymphocytic leukemia; SRPBL, splenic dif fuse red pulp small B-cell lymphoma; NHL, non-Hodgkin ’s lymphoma; HL, Hodgkin lymphoma; SLL, small lymphocytic lymphoma; DLBL, dif fuse large B-cell lymphoma; ABC, activated B cell-like; GCB, center B cell-like; MM, multiple myeloma; PBL, plasmablastic lymphoma; PB, peripheral blood; BM, bone marrow; LN, lymph node; CE, cavity exudate; FNA, fine needle aspiration; CSF, cerebrospinal fluid; IHC, immunohistochemistry, MFI, mean fluorescence intensity. *Three cases of MCL moderate or bright CD200 expression (all three cases showed cyclin CD1 overexpression by IHC; two cases had t(11;14)(q12:q32) by F ISH and conventional cytogenetics also; one case showed del(14q31q32) in three of 20 metaphases. Flow cytometry immunophenotyping in all three cases showed an atypical pattern of CD23 expression, positive on most lymphoma cells). **11 cases were atypical-CLL (CD23 absent or dimly expressed in six cases and the following antigens expressed with moderate/strong intensity: FMC-7 in eight cases; CD79b in five cases, and smIg in four cases). ***CD5+ NHL (non-CLL, non-MCL). #CD200 expression was studied by means of immunohistochemistry (HIC). °mostly nodal or MALT types. °°90 FL cases were studied and a spectrum of CD200 expression ranging from negative to moderate was found (not reported the number of positive cases); o nly three LPL cases were studied with moderate expression of CD200. §56 CLL of which 13 CLL-like MBL; 7 patients with CLL were atypical. Both typical and atypical were found all CD200 positive; MFIand percentage of CD200 e xpression were found not dif ferent; no dif ferences were found between typical and atypical CLL also. çNo dif ference between typical and atypical CLL. Multivariate analysis for MCL and atypical CLL discrimination, it was demonstrated that the most det erminant molecule was CD200 (p<0.0001, 95% CI). *MFI >1,000 was used to represent positive staining while MFI ≤ 1,000 to represent negative staining for CD200. ^CD200, added to the canonical 5 markers, improved the diagnostic accuracy of Matutes score from 86.7% to 92.5% (p<.01).
D’Arena et al. CD200 and CLL
CLL from CLPDs other than MCL. These
findings suggest that
assessment of CD200 within scores rather than as a single
marker, as reported by some authors, would be more useful
(
66
–
68
).
In addition, some authors investigated whether a different
expression of CD200 between CLL and monoclonal B-cell
lymphocytosis (MBL) exists. Sorigue et al demonstrated that all
cases of both CLL and CLL-like MBL were CD200 positive.
However, MBL cases showed a lower CD200 MFI than CLL.
Moreover, both CLL with trisomy 12 and CLL-like MBL
displayed lower CD200 MFI than CLL with other cytogenetic
abnormalities. In contrast, Rawstron et al did not
find any
difference in expression of a large body of markers, including
CD200, between CLL-type MBL and CLL (
72
). However,
differences in the expression of other markers were observed,
including lower expression of CD38, CD62L, and CD49d and
higher expression of LAIR-1, CXCR5, and CCR6 on CLL-type
MBL compared to CLL. In conclusion, despite CLL-type MBL
being phenotypically identical to CLL for a large body of
antigens, certain differences exist, particularly with respect to
proteins involved in the homing to lymphoid tissue.
CD200 AND PROGNOSIS IN CLL
Little is known about the prognostic significance of CD200
expression in CLL. As a matter of fact, con
flicting results have
been reported so far. Few data have been published and both
percentage of CD200 positivity and
fluorescence intensity have
been used to estimate CD200 expression on CLL
B-lymphocytes. Firstly, Wang et al in 2014 identi
fied two
distinct groups of CLL patients according to the expression of
CD200 on bone marrow B cells (CD200 low group: <50%;
CD200 high group:
≥ 50%) (
73
). As reported in
Table 3
,
correlations were found with some clinical-biological features
of CLL patients with <50% CD200+ cells. In contrast, using the
same methodological approach, El Din Fouad et al found other
correlations in patients with a lower percentage of CD200+ CLL
cells (<50%) (
74
). Moreover, while the former study did not
report data on TTT, response to therapy or OS, the latter found
no correlation with response to treatment or OS. In a large
cohort of patients with CLL, Miao et al, evaluating the CD200
mean
fluorescence intensity (MFI) instead of the percentage of
positivity, found that patients with lower CD200 MFI had a
significantly shorter TTT with respect to patients with higher
CD200 MFI (
75
). However, no correlation was found between
CD200 MFI and OS, and CD200 MFI did not maintain its
predictive value on TTT in multivariate analysis. More recently,
using a more standardized
flow cytometric approach, we
measured the CD200 MFI on CD19+ and CD19- lymphocyte
subpopulations, calculating the relative
fluorescence intensity
(RFI) as a ratio of the CD200 MFI on CD19+ and CD19- cells
(
76
). Lower and higher CD200 RFI values were found to be
associated with del11q and del13q14, respectively. In addition,
FIGURE 3 | Overview of the study by Sorigue et al. (70). The algorithm of the study using the three diagnostic scores to assign a diagnostic category is explained in the text (58,66,69).D’Arena et al. CD200 and CLL
CD200 RFI greater than the mean cohort value was detected in
patients with longer OS. However, these data have to be
confirmed in patients with a longer follow-up.
In summary, con
flicting results have been reported on the
prognostic role of CD200 expression in CLL so far.
Methodological and sampling differences, such as the analysis
of percentage of positive cells versus MFI, bone marrow versus
peripheral blood cells, and different
flow cytometry instrument
settings and/or gating strategy, could account for these
differences. Further studies are needed to better understand
this issue before reaching definitive conclusions.
CONCLUSIONS AND FUTURE
DIRECTIONS
CD200 expression is useful in better classifying B-cell CLPDs.
The addition of CD200 to
flow cytometry marker panels
addressing the diagnosis of this heterogenous group of B cell
neoplasms may be particularly helpful in distinguishing some
disease entities, in particular CLL and MCL, whose clinical
behavior and prognosis are quite different. On the contrary,
expression of CD200 does not appear to have a relevant role as a
prognostic indicator in CLL according to data published so far,
despite limited in number. However, this issue needs to be better
addressed by studies on a larger cohort of patients and using
standardized methodologies. Finally, the potential role of CD200
as a therapeutic target must be taken into account in particular
for diseases that highly express CD200, such as CLL and
HCL (
24
,
77
). In fact, CD200 is known to act as an
immunosuppressive molecule that is upregulated on primary
CLL B-cells (
18
). Moreover, an elegant work by Kretz-Rommel
and co-workers demonstrated that CD200 expression by tumor
cells suppresses antitumor responses in an animal model (
22
). In
a similar fashion, Gorczynski et al showed that by manipulating
CD200:CD200R interactions it is possible to cure local tumors
and distant metastases in a murine breast cancer model (
78
,
79
).
Very recently, the
first-in-human study investigating the
therapeutic use of the recombinant humanized monoclonal
anti-CD200 antibody samalizumab in 23 patients with
advanced CLL and 3 patients with MM was published (phase 1
study NCT00648738) (
80
). Treatment was associated with mild
to moderate adverse events and resulted in a dose-dependent
decrease in CD200 expression on CLL cells. Decreased tumor
burden was also observed in 14 CLL patients, with one of them
achieving a durable partial response, while 16 patients maintained
a stable disease. These results, although preliminary, suggest that
samalizumab could represent an immune checkpoint inhibitor
with activity in CLL.
Finally, there is some evidence that serum levels of soluble
CD200 may be related to disease progression and prognosis in
patients with CLL (
79
,
81
,
82
). In particular, Wong et al showed
that CD200 can be released from CD200+ neoplastic cells by
ectodomain shedding (
81
). This event is regulated by ADAM28
(
79
,
82
). Interestingly, both the membrane and the soluble form
of CD200 is able to engage CD200R, which in turn can result in
increased tumor growth (
20
,
83
). This happens by means of
reduction of immune reactivity and/or increment of induction/
activation of regulatory T cells in the tumor microenvironment
(
83
,
84
). Taken together, more and more convincing data have
been reported in the literature from which the relevant role of
CD200 is emerging not only as a diagnostic and prognostic tool
but also as a potential therapeutic target in various neoplasms
including CLL.
AUTHOR CONTRIBUTIONS
GD
’A, GP, VD, FA, SD, DE, AS, and LL designed the project,
revised the scienti
fic literature, and wrote the paper. GD’A, GP,
ES, GM, and OV followed patients with chronic lymphocytic
leukemia. FL, FD’A, TS, and LV performed flow cytometric
studies. All authors contributed to the article and approved the
submitted version.
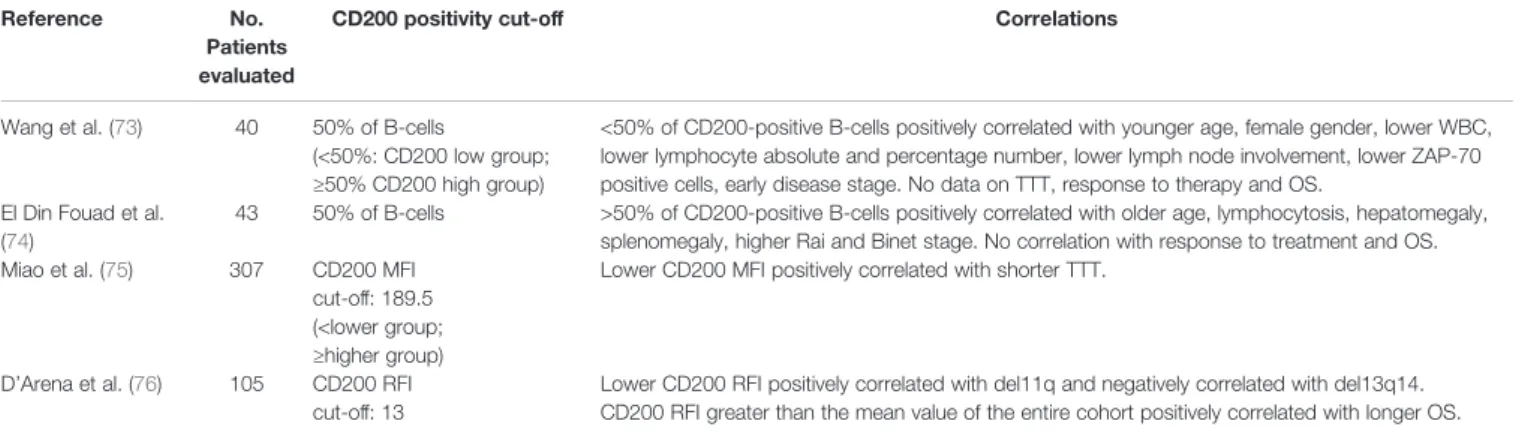
TABLE 3 | CD200 expression and prognosis in CLL.
Reference No.
Patients evaluated
CD200 positivity cut-off Correlations
Wang et al. (73) 40 50% of B-cells
(<50%: CD200 low group; ≥50% CD200 high group)
<50% of CD200-positive B-cells positively correlated with younger age, female gender, lower WBC, lower lymphocyte absolute and percentage number, lower lymph node involvement, lower ZAP-70 positive cells, early disease stage. No data on TTT, response to therapy and OS.
El Din Fouad et al. (74)
43 50% of B-cells >50% of CD200-positive B-cells positively correlated with older age, lymphocytosis, hepatomegaly, splenomegaly, higher Rai and Binet stage. No correlation with response to treatment and OS. Miao et al. (75) 307 CD200 MFI
cut-off: 189.5 (<lower group; ≥higher group)
Lower CD200 MFI positively correlated with shorter TTT.
D’Arena et al. (76) 105 CD200 RFI cut-off: 13
Lower CD200 RFI positively correlated with del11q and negatively correlated with del13q14. CD200 RFI greater than the mean value of the entire cohort positively correlated with longer OS.
WBC, white blood cell count; TTT, time to treatment; OS, overall survival; MFI, meanfluorescence intensity; RFI, relative fluorescence intensity (ratio of MFI of CD200 on CD19+ lymphocytes/MFI of CD200 on CD19- lymphocytes).
D’Arena et al. CD200 and CLL
REFERENCES
1. McCaughan GW, Clark MJ, Hurst J, Grosveld F, Barclay AN. The gene for MRC OX-2 membrane glycoprotein is localised on human chromosome 3. Immunogenetics (1987) 25:133–5. doi: 10.1007/BF00364281
2. Mathews L, Farrar W. CD200 (CD200 molecule). Atlas Genet. Cytogenet.
Oncol Hematol (2009) 13:793–8.
3. Barclay AN, Clark MJ, McCaughan GW. Neuronal/lymphoid membrane glycoprotein MRC OX-2 is a member of the immunoglobulin superfamily with a light-chain-like structure. Biochem Soc Symp (1986) 51:149–57. 4. Douglas J, Albertson DG, Barclay AN, Davis M, Rabbits PH. 2RFLP and
mapping of human MOX-1 gene on chromosome 3. Nucleic Acids Res (1988) 16:9067. doi: 10.1093/nar/16.18.9067
5. Wright GJ, Puklavec MJ, Willis AC, Hock RM, Sedgwick JD, Brown MH, et al. Lymphoid/neuronal cell surface OX2 glycoprotein recognizes a novel receptor on macrophages implicated in the control of their function. Immunity (2000) 13:233–42. doi: 10.4049/jimmunol.171.6.3034
6. Wright GJ, Jones M, Puklavec MJ, Brown JH, Barclay AN. The unusual distribution of the neuronal/lymphoid cell surface C200 (OX2) glycoprotein is conserved in humans. Immunology (2001) 102:173–9. doi: 10.1046/j1365-2567.2001.01163.x
7. Wright GJ, Cherwinski H, Foster-Cuevas M, Brooke G, Puklavec MJ, Bigler M, et al. Characterization of the CD200 receptor family in mice and humans and their interactions with CD200. J Immunol (2003) 171:3024–46. doi: 10.4049/jimmunol.171.6.3034
8. Kawasaki BT, Farrar WL. Cancer stem Cells, CD200 and immunoevasion.
Trends Immunol (2008) 29:464–8. doi: 10.1016/j.it.2008.07.005
9. Gorczynski R, Khatri I, Lee L, Boudakov I. An interaction between CD200 and monoclonal antibody agonists to CD200R2 in development of dendritic cells that preferentially induce populations of CD4+CD25+ T regulatory cells.
J Immunol (2008) 180:5946–55. doi: 10.4049/jimmunol.180.9.5946
10. Gorczynski RM, Lee L, Boudakov I. Augmented induction of CD4+CD25+ Treg using monoclonal antibodies to CD200R. Transplantation (2005) 79:488–91.
11. Zhang S, Cherwinski H, Sedgwick JD, Phillips JH. Molecular mechanisms of CD200 inhibition of mast cell activation. J Immunol (2004) 173:6786–93. doi: 10.4049/jimmunol.173.11.6786
12. Cherwinski HM, Murphy CA, Joyce BL, Bigler ME, Song YS, Zurawski SM, et al. The CD200 receptor is a novel and potent regulator of murine and human mast cell function. J Immunol (2005) 174:1348–56. doi: 10.4049/ jimmunol.174.3.1348
13. Shiratori I, Yamaguchi M, Suzukawa M, Yamamoto K, Lanier LL, Saito T, et al. Down-regulation of basophil function by human CD200 and human herpesvirus-8 CD200. J Immunol (2005) 175:4441–9. doi: 10.4049/ jimmunol.175.7.4441
14. Coles SJ, Wang ECY, Man S, Hills RK, Burnett AK, Tonks A, et al. CD200 expression suppresses natural killer cell function and directly inhibits patient anti-tumor response in acute myeloid leukemia. Leukemia (2011) 25:792–9. doi: 10.1038/leu.2011.1
15. Hoek RM, Ruuls SR, Murphy CA, Wright GJ, Goddard R, Zurawski SM, et al. Down-regulation of the macrophage lineage through interaction with OX2 (CD200). Science (2000) 290:1768–71. doi: 10.1126/science.290. 5497.1768
16. Gorczynski L, Chen Z, Hu J, Kai Y, Lei V, Ramakrishna V, et al. Evidence that an OX2-positive cell can inhibit the stimulation of type1 cytokine production by bone marrow-derived B7-1 (and B7-2)-positive dendritic cells. J Immunol (1999) 162:774–81.
17. Gorczynski RM. Transplant tolerance modifying antibody to CD200 receptor,
but not CD200, alters cytokine production profile from stimulated
macrophages. Eur J Immunol (2001) 31:2331–7. doi: 10.1002/1521-4141 (200108)31:8<2331::aid-immu2331>3.0.c0:2-#
18. McWhirter JRA, Kretz-Rommel A, Saven T, Maruyama T, Potter KN, Mockridge CI, et al. Antibodies selected from combinatorial libraries block a tumor antigen that plays a key role in immunomodulation. Proc Natl Acad Sci USA (2006) 103:1041–6. doi: 10.1073/pnas.0510081103
19. Moreaux J, Veyrune JL, Reme T, De Vos J, Klein B. CD200: a putative
theraputic target in cancer. Biochem Biophys Res Commun (2008) 366:117–22.
doi: 10.1016/j.bbrc.2007.11.103
20. Moreaux J, Hose D, Reme T, Jourdan E, Hundemer M, Legouffe E, et al. CD200 is a new prognostic factor in multiple myeloma. Blood (2006) 108:4194–7. doi: 10.118/blood-2006-06-029355
21. Tonks A, Hills R, White P, Rosie B, Mills KI, Burnett AK, et al. CD200 as a prognostic factor in acute myeloid leukaemia. Leukemia (2007) 21:566–8. doi: 10.1038/sj.leu.2404559
22. Kretz-Rommel AF, Qin N, Dakappagari EP, Ravey EP, McWhirter J, Oltean D, et al. CD200 expression on tumor cells suppresses antitumor immunity: new approaches to cancer immunotherapy. J Immunol (2007) 178:5595–602. doi: 10.4049/jimmunol.178.9.5595
23. Kretz-Rommel A, Bowdish KS. Rationale for anti-CD200 immunotherapy in B-CLL and other hematologic malignancies: new concepts in blocking immune suppression. Expert Opin Biol Ther (2008) 8:5–15. doi: 10.1517/ 14712598.8.1.5
24. Kretz-Rommel A, Qin F, Dakappagari N, Cofiell R, Faas SJ, Bowdish KS. Blockade of CD200 in the presence or absence of antibody effector function: implications for anti-CD200 therapy. J Immunol (2008) 180:699–705. doi: 10.4049/jimmunol.180.2.699
25. Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, et al. WHO classification of tumours of haematopoietic and lymphoid tissues. In: FT Bosman, ES Jaffe, SR Lakhani, H Ohgaki, editors. World Health Organization
Classification of Tumours. Lyon, France: IARC (2008).
26. Swerdlow SH, Campo E, Pileri SA, Harris NL, Stein H, Siebert R, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood (2016) 127:2375–90. doi: 10.1182/blood-2016-01-643569
27. D’Arena G, Keating MJ, Carotenuto M. Chronic lymphoproliferative
disorders: an integrated point of view for the differential diagnosis. Leuk Lymphoma (2000) 36(3-4):225–37. doi: 10.3109/1042819000914884 2000. 28. Matutes E, Owusu-Ankomah K, Morilla R, Garcia Marco J, Houlihan A, Que
TH, et al. The immunological profile of B-cell disorders and proposal of a
scoring system for the diagnosis of CLL. Leukemia (1994) 8:1640–5. 29. Moreau E, Matutes E, A’Hern RP, Morilla AM, Owusu-Ankomah A, Seon BK,
et al. Improvement of the chronic lymphocytic leukemia scoring system with
the monoclonal antibody SN8 (CD79b). Am J Clin Pathol (1997) 108:378–82.
doi: 10.1093/ajcp/108.4.378
30. Brunetti L, Di Noto R, Abate G, Gorrese A, Gravetti A, Raia M, et al. CD200/ OX2, a cell surface molecule with immuno-regulatory function is consistently expressed on hairy cell leukaemia neoplastic cells. Br J Haematol (2009) 145:665–7. doi: 10.1111/j.1365-2141.2009.07644.x
31. Palumbo GA, Parrinello N, Fargione G, Cardillo K, Chiarenza A, Berretta S, et al. CD200 expression may help in differential diagnosis between mantle cell lymphoma and B-cell chronic lymphocytic leukemia. Leuk Res (2009) 33:1212–6. doi: 10.1016/j.leukeres.2009.01.017
32. Dorfman DM, Shahsafaei A. CD200 (OX-2 membrane glycoprotein) expression in B cell-derived neoplasms. Am J Clin Pathol (2010) 134:726– 33. doi: 10.1309/AJCP38XRRUGSQOVc
33. Bhatnagar N, Wright F, McAleese D, McNamara CJ. The utility of CD200 expression in the differentiation on mantle cell lymphoma and B-cell chronic lymphocytic leukemia: a single centre experience. Blood (2010) 116(21):702. doi: 10.1182/blood.V116.21.1673.1673
34. El Desoukey NA, Afify RA, Amin DG, Mohammed RF. CD200 expression in B-cell chronic lymphoproliferatve disorders. J Invest Med (2012) 60:56–61. doi: 10.2310/JIM.0b13e31823908fb
35. Cherian S, Bohling S, Wilson M, Levin G, Wood B. Utility of CD200 in the diagnosis of chronic lymphocytic leukemia. Cytometry Part B- Clin Cytom (2012) 82B(5):329.
36. Kern W, Schabath R, Haferlach C, Schnittger S, Haferlach T. Diagnostic value of CD200 expression for the differentiation between chronic lymphocytic leukemia (CLL), CLL with increased prolymphocytes (CLL/PL) and mantle cell lymphoma. Blood (2012) 120(21):2660. doi: 10.1182/blood.V120.21. 2660.2660
37. Alapat D, Coviello-Maile JM, Owens R, Qu P, Barlogie B, Shaughnessy JD, et al. Diagnostic usefulness and prognostic impact of CD200 expression in lymphoid malignancies and plasma cell myeloma. Am J Clin Pathol (2012) 137:93–100. doi: 10.1309/AJCP59UORCYZEVQO
38. Pillai V, Pozdnyakova O, Charest K, Li B, Shahsafaei A, Dorfman DM. CD200 flow cytometric assessment and semiquantitative immunohistochemical staining distinguishes hairy cell leukemia from hairy cell leukemia-variant
D’Arena et al. CD200 and CLL
and other B-cell lymphoproliferative disorders. Am J Clin Pathol (2013) 140:536–43. doi: 10.1309/AJCPEBK31VQQNDDR
39. Dasgupta A, Ramani M, Mehrotra V, Parab S, Yadav J, Gujarathi P. CD200
expression in B-chronic lymphocytic leukemia– it is not an all or none
phenomenon. Cytometry Part B- Clin Cytom (2013) 84(6):406.
40. El-Sewefy DA, Khattab DA, Sallam MTH, Elsalakawy WA. Flow cytometric evaluation of CD200 as a tool for differentiation between chronic lymphocytic leukemia and mantle cell lymphoma. Egyptian J Haematol (2014) 39:42–6. 41. Sandes AF, Chauffaille ML, Oliveira CRMC, Maekawa Y, Tamashiro N, Takao
TT, et al. CD200 has an important role in the differential diagnosis of mature
B-cell neoplasms by multioparameterflow cytometry. Cytometry B Clin Citom
(2014) 86:98–105. doi: 10.1002/cyto.b.21128
42. Challagundla P, Medeiros LJ, Kanagal-Shamanna R, Miranda RN, Jorgensen
JL. Differential expression of CD200 in B-cell neoplasms byflow cytometry
can assist in diagnosis, subclassification, and bone marrow staging. Amer J Clin Pathol (2014) 142:837–44. doi: 10.1309/AJCPBV9ELXCOECVL 43. Karban J, Radek M, Babunkova E, Kvasnicka J, Jaksa R, Berkova A, et al.
CD200 expression improves differential diagnosis between chronic lymphocytic leukemia and mantle cell lymphoma. Blood (2014) 124 (21):5637. doi: 10.1182/blood.V124.21.5637.5637
44. Sorigue M, Junca J, Grananda I. CD200 in high-grade lymphoma, chronic lymphocytic leukemia, and chronic lymphocytic leukemia-phenotype monoclonal B-cell lymphocytosis. Am J Clin Pathol (2015) 144:676–9. doi: 10.1309/AJCP02KKFMOEXNZF
45. Lesesve J-F, Tardy S, Frotscher B, Latger-Cannard V, Feugier P, De Carvalho M, et al. Combination of CD160 and CD200 as a useful tool for differential diagnosis between chronic lymphocytic leukemia and other mature B-cell neoplasms. Int J Lab Hematol (2015) 37:486–94. doi: 10.1111/ijlh.12315
46. Fan L, Miao Y, Yu-Jie W, Wang J, Guo R, Wang L, et al. Expression patterns of CD200 and CD148 in leukemic B-cell chronic lymphoprolifeartive disorders and their potential value in differential diagnosis. Leuk Lymphoma (2015)
56:3329–35. doi: 10.3109/10428194.2015.1030642
47. Naseem S, Poongodi R, Varma N, Malhotra P, Varma S. Utility of CD200 expression and CD20 antibody binding capacity in differentiating chronic lymphocytic leukemia from other chronic lymphoproliferative disorders. Blood (2015) 126(23):5278. doi: 10.1182/blood.V126.23.5278.5278 48. Rahman K, Kumari S, Singh MK, Gupta R, Yadov G, Kumari N, et al. Atypical
presentation of hairy cell leukemia: significance of CD200 on flow cytometry. J Canc Res Ther (2018) 14:1130–4. doi: 10.4103/0974-1482.188432 49. Rahman K, Kumar K, Gupta R, Singh MK, Nityanand S. Role of CD200 in
differential diagnosis of mature B-cell neoplasms. Int J Lab Hematol (2017) 39:384–91. doi: 10.111/ijlh.12637
50. Ting YS, Smith SABC, Brown DA, Dodds AJ, Fay KC, Ma DDF, et al. CD200 is a useful diagnostic marker for identifying atypical chronic lymhocytic leukemia
byflow cytometry. Int J Lab Hematol (2018) 40(5):533–9. doi: 10.1111/ijlh.12857
May 27.
51. Arlindo EM, Marcondes NA, Fernandes FB, Faulhaber GAM. Quantitative flow cytometric evaluation of CD200, CD123, CD43 and CD52 as a tool for the differential diagnosis of mature B-cell neoplasms. Rev Bras Hematol Hemother (2017) 39:252–8. doi: 10.1016/j.bjhh-2017.05.002
52. Mason EF, Pzdnyakova O, Li B, Dudley G, Dorfman DM. Flow cytometric patterns of CD200 and cD1d expression distinguish CD10-negative, CD5-negative mature B-cell lymphoproliferative disorders. Am J Clin Pathol (2017) 148:33–41. doi: 10.1093/AJCP/AQX041
53. Starostka D, Kriegova E, Kudelka M, Mikula P, Zehnalova S, Radvansky M, et al. Quantitative assessment of informative immunophenotypic markers increases the diagostic value of immunophenotyping in mature CD5-positive
B-cell neoplasms. Cytometry Part B– Clin Cytometry (2017) 94B:576–87. doi:
10.1002/cyto.b.21607
54. Miao Y, Cao L, Sun Q, Li X-T, Wang Y, Qiao C, et al. Spectum and immunophenotyping of 653 patients with B-cell chronic lymphoproliferative disorders in China: a single-centre analysis. Hematol Oncol (2018) 36:121–7. doi: 10.1002/hon.2461
55. Poongodi R, Varma N, Naseem S, Parveen B, Varma S. Utility of CD200 expression and CD20 antibody binding capacity in differentiating chronic lymphocytic leukemia from other chronic lymphoproliferative disorders. Indian J Pathol Microb (2018) 61:50–7. doi: 10.4103/IJPM.IJPM_267_17
56. Favre R, Manzoni D, Traverse-Glehen A, Verney A, Jallades L, Callet-Bauchu E, et al. Usefulness of CD200 in the differential diagnosis of SDRPL, SMZL, and HCL. Int J Lab Hem (2018) 40:e59–62. doi: 10.1111/ijlh.12824 57. Falay M, Ozturk BA, Gunes K, Kalpakci Y, Dgdas S, Ceran F, et al. The role of
CD200 and CD43 expression in differential diagnosis between chronic lymphocytic leukemia and mantle cell lymphoma. Turk J Hematol (2018) 35:94–8. doi: 10.4274/tih.2017.0085
58. D’Arena G, Vitale C, Rossi G, Coscia M, Omedè P, D’Auria F, et al. CD200
included in a 4-marker modified Matutes score provides optimal sensitivity
and specificity for the diagnosis of chronic lymphocytic leukaemia. Hematol Oncol (2018) 36:543–6. doi: 10.1002/hon.2510
59. Debord C, Robillard N, Theisen O, Gastinne T, Godmer P, Graveleau J, et al.
CD200 expression in flow cytometry helps to distinguish mantle cell
lymphoma from other CD5-positive B-cell neoplasms. Hematol Oncol (2018) 36:307–609. doi: 10.1002/hon.2511
60. Mora A, Bosch R, Cuellar C, Vicente EP, Blanco R, Martino JM, et al. CD200 is a useful marker in the diagnosis of chronic lymphocytic leukemia. Cytometry B Clin Cytom (2019) 96:143–8. doi: 10.1002/ cyto.b.217222
61. Myles N, Giri P, Chim I, Kodituwakku A. The utility of CD200 expression and
modified Matutes score in the diagnostic differentiation of mantle cell
lymhoma and chronic lymphocytic leukemia using flow cytometry. Leuk
Lymphoma Sep (2019) 27:1–3. doi: 10.1080/10428194.2019.1663420 62. Soong N, Das-Dupta E, Dearden A. Evaluation of the diagnostic usefulness
of CD200 marker in chronic lymphocytic leukaemia and mature B-cell lymphoproliferative disorders. Br J Haematol 2019 (2019) 185(Suppl. 1):87. 63. El-Neanaey WA, Swelem RS, Ghallab OM, Abu-Shelou SM. Evaluation of
CD160 and CD200 expression as differentiating markers between chronic lymphocytic leukemia and other mature B-cell neoplasms. Int J Hematol Oncol Stem Cell Res (2020) 14:27–37.
64. Anumanthan A, Bensussan A, Boumsell L, Christ AD, Blumberg RS, Voss SD, et al. Cloning of BY55, a novel Ig superfamily member expressed on NK cells, CTL, and intestinal intraepithelial lymphocytes. J Immunol (1998) 161:2780–90.
65. Liu FT, Giustiniani J, Farren T, Tia L, Bensussan A, Gribben JG, et al. CD160 signaling mediates PI3K-dependent survival and growth signals in chronic
lymphocytic leukemia. Blood (2010) 115:3079–88. doi:
10.1182/blood-2009-08-239483
66. Khonke T, Wittmann VK, Bucklein VL, Lichtenegger F, Pasalic Z, Hiddemann W, et al. Diagnosis of CLL revisited: increased specificity by a
modified five-marker scoring system including CD200. Br J Haematol (2017)
179:480–7. doi: 10.1111/bjh.14901
67. Sorigue M, Franch-Sarto M, Sarrate E, Juca J. Usefulness of the CLLflow score. Cytometry B Clin Cytom (2018) 94(6):950–2. doi: 10.1002/cyto.b.21623
68. D’Arena G, Vitale C, Coscia M, D’Auria F, Bellesi G, Topini G, et al. External
validation of the accuracy of‘CLLflow score’. J Investig Med (2018) 66(e):e6.
doi: 10.1136/jim-2018-000832
69. Sorigue M, Junca J, Sarrate E, Grau J. Expression of CD43 in chronic
lymphoproliferative leukemias. Cytometry B Clin Cytom (2018) 94:136–42.
doi: 10.1002/cyto.b.21509
70. Sorigue M, Raya M, Vergara S, Serrate E, Orna E, Junca J. Refining the limits of borderline lymphoproliferative disorders. Cytometry B Clin Cytom (2018)
96:351–8. doi: 10.1002/cyto.b.21760
71. Sorigue M, Magnano L, Miljkovic MD, Nieto-Moragas J, Santos-Gomez M, Villamor N, et al. Positive predictive value of CD200 positivity in the differential diagnosis of chronic lymphocytic leukemia. Cytometry (2019) 98
(5):441–8. doi: 10.1002/cyto.b.21849
72. Rawstron AC, Shingles J, de Tute R, Bennett F, Jack AS, Hillmenn P. Chronic lymphocytic leukemia (CLL) and CLL-type monoclonal B-cell lymphocytosis (MBL) show differential expression of molecules involved in lymphoid tissue
homing. Cytometry B Clin Cytom (2010) 78B(Suppl 1):S42–6. doi: 10.1002/
cyto.b.20534
73. Wang X, Zhang Z, Liu Y, Wang L, Yuan H, Xie P, et al. Expression of CD200 in the bone marrow of chronic lymphocytic leukemia patients and its correlations with clinical prognosis. Chin J Cell Mol Immunol (2014) 30:75–8.
74. El Din Fouad NB, Ibrahim NY, Abdel Aziz RS, Ibrahim SK. CD200 expression in diagnostic and prognostic assessment of mature B cell lymphoproliferative
D’Arena et al. CD200 and CLL
neoplasms. Asian Pac J Cancer Prev (2018) 19:3383–92. doi: 10.31557/ APJCP.2018.19.12.338
75. Miao Y, Fan L, Wu Y-J, Xia Y, Qiao C, Wang Y, et al. Low expression of CD200 predicts shorter time-to treatment in chronic lymphocytic leukemia.
Oncotarget (2016) 7:13551–62. doi: 10.18632/oncotarget.6948
76. D’Arena G, Valvano L, Vitale C, Coscia M, Statuto T, Bellesi S, et al. CD200 and prognosis in chronic lymphocytic leukemia: conflicting results. Leuk Res (2019) 83:106169. doi: 10.1016/j.leukres.2019.106169
77. Ljungars A, Martensson L, Mattson J, Kovacek M, Sundberg A, Tornberg UC, et al. A platform for phenotypic discovery of therapeutic antibodies and targets applied on chronic lymphocytic leukemia. Precis Oncol (2018) 2:18. doi: 10.1038/s41698-018-0061-2
78. Gorczynski RM, Chen Z, Khatri I, Podnos A, Yu K. Cure of metastatic growth of EMT6 tumor cells in mice following manipulation of CD200:CD200R signaling. Breast Cancer Res Treat (2013) 142:271–82. doi: 10.1007/s10549-013-2735-3
79. Wong KK, Zhu F, Khatri I, Huo Q, Spaner DE, Gorczynski RM. Characterization of CD200 ectodomain shedding. PloS One 2016; (2016) 11:e0152073. doi: 10.1371/journal.pone.0152073
80. Mahadevan D, Lanasa MC, Farber C, Pandey M, Whelden M, Faas SJ, et al. Phase I study of samalizumab in chronic lymphocytic leukemia and multiple myeloma: blockade of the immune checkpoint CD200. J ImmunoTher Canc 7 (2019) 227:1–13. doi: 10.1186/s40425-019-0710-1
81. Wong KK, Brenneman F, Chesney A, Spaner DE, Gorczynski RM. Soluble CD200 is critical to engraft chronic lymphocytic leukemia cells in
immunocompromised mice. Cancer Res (2012) 72:4931–43. doi: 10.1158/
0008-5472.CAN-12-1390
82. Twito T, Chen Z, Khatri I, Wong K, Spaner D, Gorczynski RM. Ectodomain shedding of CD200 from the B-CLL surface is regulated by ADAM28
expression. Leuk Res (2013) 37:816–21. doi: 10.1016/j.leukres.2013.04.014
83. Norde WJ, Hobo W, van der Voort R, Dolstra H. Coinhibitory molecules in hematologic malignancies: targets for therapeutic intervention. Blood (2012) 120:728–36. doi: 10.1182/blood-2012-02-412510
84. Jadidi-Niaragh F, Yousefi M, Memarian A, Hojjat-Farsangi M, Khoshnoodi J,
Razavi SM, et al. Increased frequency of CD8+ and CD4+ regulatory T cells in chronic lymphocytic leukemia: association with disease progression. Cancer Invest (2013) 31:121–31. doi: 10.3109/07357907.2012.756110
Conflict of Interest: The authors declare that the research was conducted in the
absence of any commercial orfinancial relationships that could be construed as a
potential conflict of interest.
Copyright © 2020 D’Arena, De Feo, Pietrantuono, Seneca, Mansueto, Villani, La Rocca,
D’Auria, Statuto, Valvano, Arruga, Deaglio, Efremov, Sgambato and Laurenti. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
D’Arena et al. CD200 and CLL