Sunitinib administered on 2/1 schedule in patients with
metastatic renal cell carcinoma: the RAINBOW analysis
S. Bracarda, R. Iacovelli, L. Boni, M. Rizzo, L. Derosa, M. Rossi, L. Galli, G. Procopio, M. Sisani,
F. Longo, M. Santoni, F. Morelli, G. Di Lorenzo, A. Altavilla, C. Porta, A. Camerini & B. Escudier
on behalf of the Rainbow Group
Ann Oncol 2015; 26: 2107
–2113 (doi: 10.1093/annonc/
mdv315)
In the original article, Figures 1 and 2 were interchanged.
The correct presentation is given below.
100 90 80 70 60 Ov er all sur viv al probability , % 50 40 30 20 10 0 0 1 2 3 4 Follow-up, yrs No. at risk* Group 4/2 to 2/1 207 39 211 172 16 141 110 5 82 57 4 50 32 1 29 14 0 20 5 0 10 1 0 5 0 0 2 0 0 1 0 0 0 Group 2/1 Controls 5 6 7 8 9 10 Group 4/2 to 2/1 Group 2/1 Controls
Figure 2. Overall survival. Asterisk denotes one patient in the 4/2→2/1 group and two patients in the 2/1 group were not evaluable for OS.
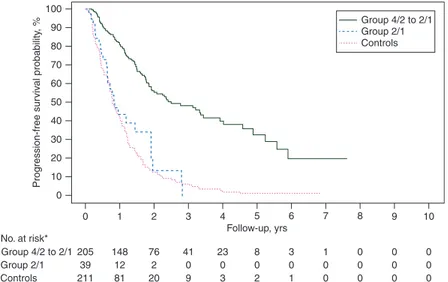
100 90 80 70 60 Prog ression-free sur viv al probability , % 50 40 30 20 10 0 0 1 2 3 4 Follow-up, yrs No. at risk* Group 4/2 to 2/1 205 39 211 148 12 81 76 2 20 41 0 9 23 0 3 8 0 2 3 0 1 1 0 0 0 0 0 0 0 0 0 0 0 Group 2/1 Controls 5 6 Group 4/2 to 2/1 Group 2/1 Controls 7 8 9 10
Figure 1. Progression-free survival. Asterisk denotes three patients in the 4/2→2/1 group and two patients in the 2/1 group were not evaluable for PFS.
corrigendum
corrigendum
Annals of Oncology 27: 366, 2016doi:10.1093/annonc/mdv589 Published online 18 December 2015
© The Author 2015. Published by Oxford University Press on behalf of the European Society for Medical Oncology. All rights reserved. For permissions, please email: [email protected].