Available
online
at
www.sciencedirect.com
journal
homepage:
www.elsevier.com/locate/radcr
Case
Report
Magnetic
resonance
enterography
appraisal
of
lupus
enteritis:
A
case
report
Giuseppe
Cicero,
MD
∗,
Alfredo
Blandino,
MD,
Tommaso
D’Angelo,
MD,
Antonio
Bottari,
MD,
Marco
Cavallaro,
MD,
Giorgio
Ascenti,
MD,
Silvio
Mazziotti,
MD
SectionofRadiologicalSciences,DepartmentofBiomedicalSciencesandMorphologicalandFunctionalImaging, UniversityofMessina,Policlinico“G.Martino” ViaConsolareValeria,1,Messina98100,Italy
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:Received27March2018 Revised8June2018 Accepted12June2018 Availableonline11July2018
Keywords:
Magneticresonanceenterography Lupusenteritis
a
b
s
t
r
a
c
t
Systemiclupuserythematosus(SLE)isachronicautoimmunediseasewithamultisystemic involvement.Usually,radiologicalimagingdoesnotplayacentralroleinevaluatingSLE patients,althoughitmaybehelpfulinassessingcomplications,allowingamoreaccurate evaluationofthepatient.Lupusenteritisisoneofthemostcommonandpotentiallylethal manifestationsofthegastrointestinalinvolvementofSLE.Amongtheimagingmodalities, computedtomographyscanisnowconsideredthegoldstandardinevaluatinglupus en-teritis,althoughitisimpairedbytheradiationexposure.Ontheotherhand,duringthelast decademagneticresonanceenterographyhasachievedaremarkableimportancein evalu-atingsmallbowellesionsinpatientsaffectedbyCrohn’sdisease.Wedescribethefirstcase reportoflupusenteritisevaluatedwithmagneticresonanceenterography,puttingforward theproposalofareliableandradiation-freealternativetocomputedtomographyscanin evaluatingtheintestinalinvolvementofSLE.
© 2018TheAuthors.PublishedbyElsevierInc.onbehalfofUniversityofWashington. ThisisanopenaccessarticleundertheCCBY-NC-NDlicense. (http://creativecommons.org/licenses/by-nc-nd/4.0/)
Introduction
Systemic lupuserythematosus (SLE) is achronic multisys-temicautoimmune disease whose specific etiology still re-mainsunknown[1,2].
Ageneticpredispositionandsomeenvironmentalrisk fac-torscontributetoitsonset,leadingtoanalteredimmune re-sponseconsistinginhyperactivationofTandBlymphocytes, lossofself-tolerance,andformationofcirculatingpathogenic
R CompetingInterests:None. ∗ Correspondingauthor.
E-mailaddress:[email protected](G.Cicero).
immune complexes, withtheir consequent deposition and damageofseveralorgans[1,2].
TheoverallincidenceratesforSLEareapproximately 0.3-23.7per 100,000person-years,withaprevalencethatrange from6.5to178.0per100,000andafemale–maleratiocloseto 9:1[2,3].
Althoughthediagnosisandtheevaluationofthediseaseas awholearestrictlyclinical,theassessmentandthefollow-up ofsomecomplicationsmayrequiretheusefulnessof radio-logicalimaging.
Inparticular,thegastrointestinalinvolvementofSLEisa potentiallyseverecomplicationofSLE[4],withanincidence thatrangefrom5.4%to40%ofthepatients[5,6];amongits possible clinical manifestations, one of the most common https://doi.org/10.1016/j.radcr.2018.06.008
1930-0433/© 2018TheAuthors.PublishedbyElsevierInc.onbehalfofUniversityofWashington.Thisisanopenaccessarticleunderthe CCBY-NC-NDlicense.(http://creativecommons.org/licenses/by-nc-nd/4.0/)
islupusenteritis,animmunocomplex-mediatedvascular in-flammationthatmayleadtothenecrosisofthevesselwalls [5,7].
Accordingtothedefinitiongivenbythe BritishIsles Lu-pusAssessmentGroupdiseaseactivityindex,lupusenteritis isintendedasa“gastrointestinalSLEinvolvementaseither vasculitisorinflammationofthesmallbowel,withsupportive imagingand/orbiopsyfindings"[8].However,inliterature, lu-pusenteritisandlupusvasculitisareoftenusedassynonyms, togetherwithotherdenominations,suchasmesenteric arteri-tis,lupusarteritis,gastrointestinalvasculitis,intra-abdominal vasculitis,andacutegastrointestinalsyndrome[5,9,10].
Uptonow,allthedifferentimagingmodalitieshavenot shown pathognomonic signs related tolupus enteritis, in-cluding computed tomography (CT) scan, that is consid-eredthegoldstandardinvestigationinspiteoftheradiation exposure.
Magnetic resonance enterography (MRE) is a radiation-safe,full comprehensiveexaminationusually indicatedfor patientsaffectedbyCrohn’sdisease(CD).
However,consideringtheincreasinglyimportancethatthis techniquehas achievedduringthe last yearsin evaluating smallbowellesions,itispossibletoconsidernewfrontiersof itsperforming.
Toourknowledge,wedescribethefirstcasereportof gas-trointestinalinvolvementofSLEevaluatedwithMRE.
Case
report
Wedescribethecaseofa22-year-oldwomanaffectedbySLE whohadbeenhospitalizedtwice,in2differenthospitals,due togastrointestinalsymptoms.
Thefirsttime,anabdominalx-rayplainradiographanda CTscanwereobtained,showingsomegas–fluidlevelswithin theilealloops,whosewallswerealsothickenedandwitha layeredaspect;somecentimetriclymphnodeswerealso visi-bleinthemesentericfat,andperihepaticandperisplenicfluid collectionswereseen.
Abiopsythroughacolonoscopicexamwasalsoperformed, which showed mucosal ulcerative lesions in the terminal ileumwithcellularinfiltration andhemorrhage fociwithin theunderlyinglayersoftheintestinalwall,allowingthe di-agnosisoflupusenteritis.
Moreover, a US examination of both kidneys and an ultrasound-guidedbiopsyofthelowerpoleoftheleftkidney werealreadyperformed,demonstratingarenalhistologyof classIVlupusnephritis.
The patient was discharged after the prescription of steroidsandimmunosuppressivetherapy.
However,theimmunosuppressivetherapywaslater sus-pendedduetotheonsetofamarkedneutropenia.
After10monthsfromthelasthospitalization,thepatient cametotheEmergencyRoomofourhospitalduetothe re-crudescenceoftheabdominalsymptomsandtheoccurrence ofvasculiticurticariawithangioedemaoftherighteyeand thesuperiorlip.
Laboratory tests showed active renal disease, with in-creasedproteinuria(3040,70mg/24h),lowcomplement frac-tionC3(61,9mg/dL),lowC4(5.29mg/dL),increasedPCR(31,
54mg/L),high velocità di eritrosedimentazione(VES) value (40mm/h),positiveelevatedanti-ds-DNAantibodies(123,60 IU/mL),positiveantinuclearantibodyat1:1600,positive anti-Roantibodies,and a normallymphocyte countwith lower CD4+and/orCD8+ratio.
Inordertoassess thecurrentstatusoftheintestinal in-volvementandinaccordancewiththeclinicians,itwas de-cidedtoperformanMRE,withtheprincipalaimofsparing thepatientanotheramountofradiations.
MRE requires the oral administration of approximately 1500mL of polyethylene glycol-water solution, starting 45 minutesbeforethebeginningoftheexam.
Afterthepatientwasplacedinsupinepositioninsidethe scanner,coronalthick-sectionT2-weightedrapidacquisition withrelaxationenhancement(RARE)acquisition,axial and coronalT2-weightedtruefastimagingwithsteady-state pre-cession(repetition time/echo time: 4.20/2.10 ms, flip angle (FA):60°),andhalf-Fourieracquisitionsingle-shotturbospin echo(repetition time/echo time: ∞/80 ms) with and with-outfatsuppressionwereperformed,togetherwith diffusion-weighted imaging (DWI) sequences, obtained on the axial planeusingadiffusionfactorbfixedat0,400,and800s/mm2.
Coronal precontrast ultrafast 3D T1-weighted gradient-echo fat-suppressed and ultrafast axial 3D T1-weighted gradient-echofat-suppressedimagesobtainedafterinjection ofgadoteratemeglumine (Dotarem)atadose of0.2mL/kg bodyweightwereacquiredat30,60,and180seconds,followed byabolusof30mLofnormalsaline.
Theexamallowedtodetectamildthickening(5mm)of severalilealloops,whosetotalextension,measuredwith dig-italcalipersfromtheileocecalvalve,amountedto38cm.
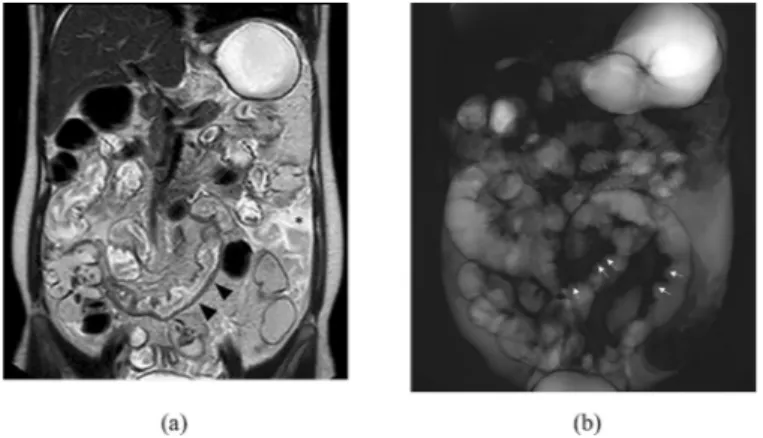
Moreover, the “thumb printing sign,” usually related to ischemic condition, was clearly detectable on T2-weighted thick-sectionRAREimages.Amoderateamountoffreefluid wasalsoseenwithintheabdominalcavity(Fig.1).
Diffusion-weighted and apparent diffusion coefficient (ADC)calculation did not show water restriction, whereas contrast-enhancedsequencesdemonstratedamild enhance-mentofthethickenedsmallbowelwalls(Fig.2).
Thepatientwastreatedwithsteroids(intravenous admin-istrationofmethylprednisolone,1g/dayfor3days)and mon-oclonalantibodies(intravenousadministrationofrituximab, 1g/day).
Afterthereliefoftheabdominalandcutaneoussymptoms andtheprescriptionofthesteroidtherapy,the patientwas discharged.
Discussion
Although several manifestations of the gastrointestinal tract involvement can be recognized in SLE patients (eg, protein-losing enteropathy, intestinal pseudo-obstruction, eosinophilicenteritis,etc.)[7],lupusenteritisremainsoneof themostcommon,affectingupto53%ofthepatients present-ingabdominalpain[4,11].
Inflammatoryenteritisisconsequenttothedepositionof circulantpathologicimmunocomplexandthrombosisofthe intestinalvessels[12];itsprevalencerangesfrom0.2%to53%
Fig. 1 – Coronal T2-weighted half-Fourier acquisition single-shot turbo-spin-echo scan (a) showing a mild thickening of a distal ileal loop ( blackarrowheads) and free intraperitoneal fluid ( asterisk). The “thumb printing sign” ( arrows) is also easily appreciable on coronal T2-weighted thick-section RARE image (b).
Fig. 2 – Coronal T1-weighted T1 high resolution isotropic volume excitation (THRIVE) with fat saturation (FS) after Gd injection sequences (a) demonstrates mild enhancement of the small bowel thickened walls ( arrows). Axial
diffusion-weighted acquisition (b) and gray-scale ADC image (c) does not show water restriction of the same pathologic loop ( arrows). S, sigma.
ofSLEpatients,althoughitisclinicallysignificantinonly2% ofthem[13].
Usually,themostaffectedtractofthegutistheone sup-pliedbythesuperiormesentericartery[6,14],inparticular je-junumandileum[5].
A wide spectrum of generic symptoms can be related tothiscondition,includingabdominalpain,fever,vomiting, anorexia,diarrhea,pancreatitis,besidessomecoexisting typ-icalsignsofSLE,suchasmalarrashorarthritis[12,13,15].
However,arapiddiagnosismayleadtoatimelytherapeutic approach:steroidsareusuallysufficient,otherwise immuno-suppressivetreatmentcanbechosenformoreseverecases [5].
Althoughthedefinitediagnosisandtheevaluationofthe diseaseentirelyremaininthehandsoftheclinicians, radi-ologicalimagingcanprovideausefulsupportinthe assess-mentandfollow-upofthiscomplication.
Nowadays, CT scan is considered the gold standard in imagingevaluationoflupusenteritis,allowingthedetection ofthetypicalfeaturesofischemicbowel:focalordiffusewall thickening,dilatationofthelumen,enhancementofthe mu-cosa and serosa (the so-called “target sign”),engorgement ofmesentericvessels(“thecombsign”),and mesentericfat stranding[5,6,12,13,15].Additionalreliefssuchasascites, lym-phadenopathies,andgenitourinaryinvolvementcanalsobe found[14].
Otherimagingmodalitiescanalsobeusedinthe evalua-tionofthiscondition:abdominalultrasoundcanbehelpful inrecognizingbowelsubmucosaledema,wallthickening,or ascites[11],whereasdouble-contrastradiographymayshow thickening andirregular profileoftheloop involveddueto hemorrhageandedema(the“thumbprinting” sign), suggest-ingbowelischemia[6,11,12].
However,allthoseradiologicalsignsarenotspecificof lu-pusenteritisandthedifferentialdiagnosismayinclude pan-creatitis,mechanicalbowelobstruction,peritonitis,or inflam-matoryboweldiseases(IBDs)[5].
Therefore,endoscopicandhistologicalconfirmationsare requiredinorder toget theright diagnosis andtoexclude otherconcomitantdiseases,althoughveryrare,suchasIBD [1,13].
Duringthe lastyears,MREhasalready beenincludedin theevaluationprotocolofCDpatients,duetoitsaccuracyin theappraisalofsmallbowellesionsandthelackofradiation exposureandinvasiveness[16,17].
Inthecasedescribed,thepatienthadalreadygota histo-logicaldiagnosisoflupusenteritis,whichwasassessedbyCT scanduringherprevioushospitalization.
Inordertore-evaluatetheintestinalinvolvementwiththe purposeofsparingherfromanotheramountofradiations,it wasdecidedtoperformanMRE.
Thisimagingmodalityallowedusthedetectionofsome findingsusuallyassociatedwithlupusenteritis,suchasawall thickeningofthelastilealloop,withaconcomitant inden-tationofthe mucosalandserosalsides,referabletoedema and/orhemorrhageofthesubmucosallayer,andamoderate amountoffluidcollectionwithintheabdominalcavity.
Althoughitiswellestablishedthatbothsmallbowel is-chemia and acute inflammatory conditions (eg, IBDs) are strictly related to hyperintensity on DWI and
contrast-enhancedimages,inourcasethesmallbowelwallsinvolved showedonlyamildcontrastenhancementandnosignificant waterrestriction.
The mostreasonablehypothesis thatcould explainthis scenariowouldbeafibroticprogressionofthe smallbowel wallsinvolved.AsalreadydescribedforIBDs,intestinal fibro-sisistypicallycharacterizedbyhypointensityonDWIandby adelayedhyperenhancementaftercontrastmediuminjection [18,19],whosedetectionwouldhaveneededatleastan addi-tionallateracquisition.However,beyondthesediscrepancies thatshouldbedeepenedwithfurtherstudies,inouropinion MREhasthepotentialtoplayacentralroleinevaluatingthe intestinalinvolvementofSLE.
Infact,fordifferentreasons,theimagingevaluationofthe smallbowelhasalwaysbeen problematicduetothe radia-tionexposure(CTscan),thepotentiallynonexhaustive evalu-ation(fluoroscopy),orthehealthcarecosts(videocapsule en-doscopy).
Through theingestionofthewatersolutionof polyethy-leneglycol(PEG),MREpermitstodistendtheintestinalloops, leadingtoanaccurateevaluationoftheirwall.
Themainadvantagesofthistechniqueconsistinthe com-prehensiveevaluationofthewholeabdominalcavity, includ-ingthepossibilityofdetectingextraintestinalfindings,andin itssafeness,whichconsenttoreperformtheexamaftershort periodsoftimeorevenafterfewminutes(ie,iftheintestinal loopsarenotwelldilated)[20].
Ofcourse,MREisalsoimpairedbysomelimitations,such asthescanningtime(about20-30minutes),theexpertiseof theradiologistandthecomplianceofthepatientinassuming theoralcontrastmedium.
Therefore,CTscanisstillconsideredtheimagingmodality ofchoiceinevaluationofacuteonsetofthiscondition,due toits widespreadavailability,thefasterscanningtime,and theoptimalimagequalityeveninpresenceofintraluminal gas,whichcouldindeedproducesusceptibilityartifactsonthe MREimages.
Moreover,MRE,aswellas theother imagingmodalities, couldnotestablishaconfidentidentificationoflupus enteri-tis,whichhastobediagnosedclinicallyandeventuallywith endoscopy.
However, on the basis of the lack of radiation and largeamountofinformationachievablewiththisexam,the benefits–costsratio seems toincline towardthe MRE tech-nique.
Obviously,furtherstudieshavetobeperformedinthisway toimprovethecurrentknowledgeandtoextendtheoutreach ofthistechniqueoutsidetheIBDborders.
Conclusion
Wedescribedthefirstcaseoflupusintestinalenteritis evalu-atedwithMRE,animagingmodalitynowmainlyperformedin patientsaffectedbyCD.Aswellastheotherimaging modal-ities,MREcannotallowmakingadefinitediagnosis but,in comparisonwiththem,itsuseisencouragedbysome advan-tages,suchasthelackofradiationandthehugeamountof informationachievable.
ProbablytherealpotentialofMREinevaluatingthesmall bowelisstillnotentirelyknownandfurtherimprovementin thissensecouldbringfuturebenefitsonpatientcare,interms ofcompletenessofdiseaseassessmentandsparingof radia-tionexposure.
R E F E R E N C E S
[1]KirbyJM, JhaveriKS, MaizlinZV, MidiaM, HaiderE, KhaliliK. Abdominalmanifestationsofsystemiclupus
erythematosus:spectrumofimagingfindings.CanAssoc RadiolJ2009;60(3):121–32.
[2]WeckerleCE, NiewoldTB.Theunexplainedfemale
predominanceofsystemiclupuserythematosus:cluesfrom geneticandcytokinestudies.ClinRevAllergyImmunol 2011;40(1):42–9.
[3]Pons-EstelGJ, Ugarte-GilMF, AlarcónGS.Epidemiologyof systemiclupuserythematosus.ExpertRevClinImmunol 2017;13(8):799–814.
[4]FortunaG, BrennanMT.Systemiclupuserythematosus: epidemiology,pathophysiology,manifestations,and management.DentClinNorthAm2013;57(4):631–55. [5]JanssensP, ArnaudL, GalicierL, MathianA, HieM, SeneD,
etal. Lupusenteritis:fromclinicalfindingstotherapeutic management.OrphanetJRareDis2013;8:67.
[6]GohYP, NaidooP, NgianGS.Imagingofsystemiclupus erythematosus.PartII:gastrointestinal,renal,and musculoskeletalmanifestations.ClinRadiol 2013;68(2):192–202.
[7]Barile-FabrisL, Hernández-CabreraMF, Barragan-GarfiasJA. Vasculitisinsystemiclupuserythematosus.CurrRheumatol Rep2014;16(9):440.
[8]IsenbergDA, RahmanA, AllenE, FarewellV, AkilM, BruceIN, etal. BILAG2004.Developmentandinitialvalidationofan updatedversionoftheBritishIslesLupusAssessment Group’sdiseaseactivityindexforpatientswithsystemic lupuserythematosus.Rheumatology(Oxford)
2005;44(7):902–6.
[9] YuanS, YeY, ChenD, QiuQ, ZhanZ, LianF, etal. Lupus mesentericvasculitis:clinicalfeaturesandassociated factorsfortherecurrenceandprognosisofdisease.Semin ArthritisRheum2014;43(6):759–66.
[10]BrewerBN, KamenDL.Gastrointestinalandhepaticdisease insystemiclupuserythematosus.RheumDisClinNorthAm 2018;44(1):165–75.
[11]EbertEC, HagspielKD.Gastrointestinalandhepatic manifestationsofsystemiclupuserythematosus.JClin Gastroenterol2011;45(5):436–41.
[12]TianX-P, ZhangX.Gastrointestinalinvolvementinsystemic lupuserythematosus:insightintopathogenesis,diagnosis andtreatment.WorldJGastroenterol2010;16(24):2971–7. [13]KatsanosKH, VoulgariPV, TsianosEV.Inflammatorybowel
diseaseandlupus:asystematicreviewoftheliterature.J CrohnsColitis2012;6(7):735–42.
[14]LalaniTA, KanneJP, HatfieldGA, ChenP.Imagingfindingsin systemiclupuserythematosus.RadioGraphics
2004;24(4):1069–86.
[15]HaHK, LeeSH, RhaSE, KimJH, ByunJY, LimHK, etal. Radiologicfeaturesofvasculitisinvolvingthe gastrointestinaltract.RadioGraphics2000;20(3):779–94. [16]MazziottiS, AscentiG, ScribanoE, GaetaM, PandolfoA, BombaciF, etal. GuidetomagneticresonanceinCrohn’s disease:fromcommonfindingstothemorerare complicances.InflammBowelDis2011;17(5):1209–22. [17]MazziottiS, BlandinoA, ScribanoE, GaetaM, MiletoA,
FriesW, etal. MRenterographyfindingsinabdominopelvic extraintestinalcomplicationsofCrohn’sdisease.JMagn ResonImaging2013;37(5):1055–63.
[18]RimolaJ, PlanellN, RodríguezS, DelgadoS, OrdásI, Ramírez-MorrosA, etal. Characterizationofinflammation andfibrosisinCrohn’sdiseaselesionsbymagnetic resonanceimaging.AmJGastroenterol2015;110(3):432–40. [19]KaushalP, SomwaruAS, CharabatyA, LevyAD.MR
enterographyofinflammatoryboweldiseasewith endoscopiccorrelation.RadioGraphics2017;37(1):116–31. [20]MazziottiS, BlandinoA.MRenterography.1sted.Berlin,