n. R1 R2 R3 R4 Resa (%) p.f. (°C) IR (nujol, cm-1) 1 H-NMR (DMSO-d6, ppm) Formula 21 CH3 (CH2)2CH3 4-NO2 H 82 151 3173, 1618, 1511, 1075, 855 0.70-0.78 (t, 3H, J=7.4, CH2CH3); 0.83-0.91 (m, 1H, CH2CH2CH3); 1.24-1.39 (m, 2H, CH2CH2CH3); 1.43-1.54 (m, 1H, CH2CH2CH3); 2.85-2.96 (m, 2H, COCONCH3); 3.06-3.14 (t, 1H, J=7.2, COCONCH3); 7.26-7.38 (m, 2H, Ar-H); 7.52-7.56 (m, 1H, Ar-H); 7.85-7.89 (m, 2H, Ar-H); 8.03-8.09 (t, 1H, J=6.5, Ar-H); 8.35-8.40 (d, 2H, J=4.4, Ar-H); 12.73 (s, 1H, NH). C20H19N3O4 Tabella 2. Costanti chimico-fisiche e caratteristiche analitiche e spettroscopiche dei derivati 21-32
N N O O R1 R2 R4 R3
n. R1 R2 R3 R4 Resa (%) p.f. (°C) IR (nujol, cm-1) 1 H-NMR (DMSO-d6, ppm) Formula 22 CH3 (CH2)2CH3 4-NO2 CH3 91 190 1613, 1511, 1065, 835, 717 0.66-0.78 (m, 3H, CH2CH3); 1.41-1.52 (m, 2H, CH2CH3); 2.51 (s, 3H, NCH3); 2.96-3.03 (t, 2H, J=7.5, NCH2CH2CH3); 3.56 (s, 3H, NCH3); 7.34-7.47 (m, 2H, Ar-H); 7.69-7.81 (m, 3H, Ar-H); 8.21-8.24 (d, 1H, J=3.1 Ar-H); 8.37-8.42 (d, 2H, J=4.3, 3Ar-H). C21H21N3O4 23 CH3 (CH2)3CH3 4-NO2 H 61 198 3173, 1726, 1608, 1516, 1075, 866, 748 0.74-0.83 (m, 3H, CH2CH2CH2CH3); 1.13-1.15 (m, 4H, CH2CH2CH2CH3); 1.39-1.55 (m, 1H, CH2CH2CH2CH3); 2.84-2.96 (m, 3H, COCONCH3); 3.09-3.16 (m, 1H, CH2CH2CH2CH3); 7.30-7.35 (m, 2H, Ar-H); 7.52-7.57 (m, 1H, Ar-Ar-H); 7.83-7.89 (m, 2H, Ar-H); 8.022-8.12 (m, 1H, Ar-H); 8.35-8.40 (m, 2H, Ar-H); 12.76 (s, 1H, NH). C21H21N3O4 24 CH3 (CH2)3CH3 4-NO2 CH3 90 195-200 1613, 1511, 1116, 1065, 845, 748 0.76-0.83 (t, 3H, J=7.1, CH2CH2CH2CH3); 1.05-1.23 (m,2H, CH2CH2CH2CH3); 1.39-1.42 (m, 2H, CH2CH2CH2CH3); 2.30 (s, 3H, COCONCH3); 2.98-3.06 (t, 2H, J=7.2, NCH2CH2CH2CH3); 3.55 (s, 3H, NCH3); 7.32-7.46 (m, 2H, Ar-H); 7.69-7.80 (m, 3H, Ar-H); 8.20-8.23 (m, 1H, Ar-H); 8.36-8.41 (m, 2H, Ar-H). C22H23N3O4 Tabella 2. continua
n. R1 R2 R3 R4 Resa (%) p.f. (°C) IR (nujol, cm-1) 1 H-NMR (DMSO-d6, ppm) Formula 25 (CH2)2CH3 (CH2)2CH3 3-OCH3 H 43 57 3173, 1576, 1105, 876, 722 0.65-0.79 (m, 6H, CH3); 1.13-1.24 (m, 2H, CH2CH2CH3); 1.407-1.484 (m, 2H, CH2CH2CH3); 2.90-3.057 (m, 4H, CH2CH2CH3); 3.825 (s, 3H, OCH3); 7.493-7.123 (m, 7H, Ar-H); 8.069-8.101 (m, 1H, Ar-H); 12.450 (s, 1H, NH). C23H26N2O3 27 (CH2)2CH3 (CH2)2CH3 4-OCH3 H 50 121-126 0.68-0.76(m, 6H, CH2CH2CH3); 1.21-1.25(m, 2H, CH2CH2CH3); 1.39-1.46(m, 2H, CH2CH2CH3); 2.74-2.93(2t, 4H, J=8 Hz, CH2CH2CH3); 3.83 (s, 3H, OCH3); 7.04-7.08(d, 2H, J=8 Hz, ArH); 7.18-7.29 (m, 2H, ArH); 7.43-7.51(m, 2H, ArH); 7.99-8.03(d, 2H, J=8 Hz, ArH); 12.34(s, 1H, NH) C23H26N2O3 28 (CH2)2CH3 (CH2)2CH3 4-OCH3 CH3 57 135 0.67-0.75 (m, 6H, CH2CH2CH3); 1.02-1.10 (m, 2H, CH2CH2CH3); 1.40-1.52 (m, 2H, CH2CH2CH3); 2.74-2.93 (2t, 4H, J=8 Hz, CH2CH2CH3); 3.50 (s, 3H, NCH3); 3.84 (s, 3H, OCH3); 7.05-7.09 (d, 2H, J=8 Hz ArH); 7.31-7.39 (m, 4H, ArH); 7.61-7.65 (d, 1H, J=8 Hz ArH); 8.19-8.23 (d, 1H, J=8Hz ArH C24H28N2O3 29 (CH2)2CH3 (CH2)2CH3 4-NH2 H 50 199-201 0.84-0.63 (m, 6H, CH2CH2CH3); 1.37-1.29 (m, 2H, CH2CH2CH3); 1.49-1.41 (m, 2H, CH2CH2CH3); 3.08-2.97 (2t, 4H, J=7 Hz, CH2CH2CH3); 5.57 (s, C22H25N3O2
2H, NH2); 6.64-6.60 (d, 2H, J=8 Hz, ArH); 7.43-7.17 (m, 5H, ArH); 7.96-7.92 (m,1H, ArH); 12.09 (s, 1H, NH) 30 (CH2)2CH3 (CH2)2CH3 4-NH2 CH3 84 178-180 O.66-0.77 (m, 6H, CH2CH2CH3); 1.11-1.23 (m, 2H, CH2CH2CH3); 1.40-1.51 (m, 2H, CH2CH2CH3); 2.72-2.90 (2t, 4H, J=8 Hz CH2CH2CH3); 3.50 (s,3H, NCH3); 7.16-7.33 (m, 6H, ArH); 7.55-7.59 (d, 1H, J=8 Hz ArH); 8.13-8.17 (d, 1H, J=8 Hz ArH); 8.51-8.61 (d, 2H, J=20 Hz NH2) C23H27N3O2 31 (CH2)2CH3 (CH2)2CH3 4-OH H 50 101-105 0.67-0.87 (m, 6H, CH2CH2CH3); 1.31-1.22 (m, 2H, CH2CH2CH3); 1.51-1.38 (m, 2H, CH2CH2CH3); 2.94-3.04 (2t, 4H, J=7 Hz, CH2CH2CH3); 4.77 (s, 1H, OH); 6.84-6.94 (m, 2H, ArH); 7.17-7.27 (m, 2H, ArH); 7.39-7.43 (m, 2H, ArH); 7.95-8.02 (m, 2H, ArH); 12.28 (s, 1H, NH) C22H24N2O3 32 (CH2)2CH3 (CH2)2CH3 4-OH CH3 42 187-189 O.67-0.77 (m, 6H, CH2CH2CH3); 1.09-1.21 (m, 2H, CH2CH2CH3); 1.40-1.51 (m, 2H, CH2CH2CH3); 2.75-2.92 (2t, 4H, J=7 Hz CH2CH2CH3); 3.50 (s,3H, NCH3); 6.85-6.89 (d, 2H, J=8 Hz ArH); 7.21-7.36 (m, 4H, ArH); 6.60-6.63 (d, 1H, J=6 Hz ArH); 8.18-8.22 (d, 1H, J=8 Hz ArH); 9.88 (s, 1H, OH) C23H26N2O3 Tabella 2. continua
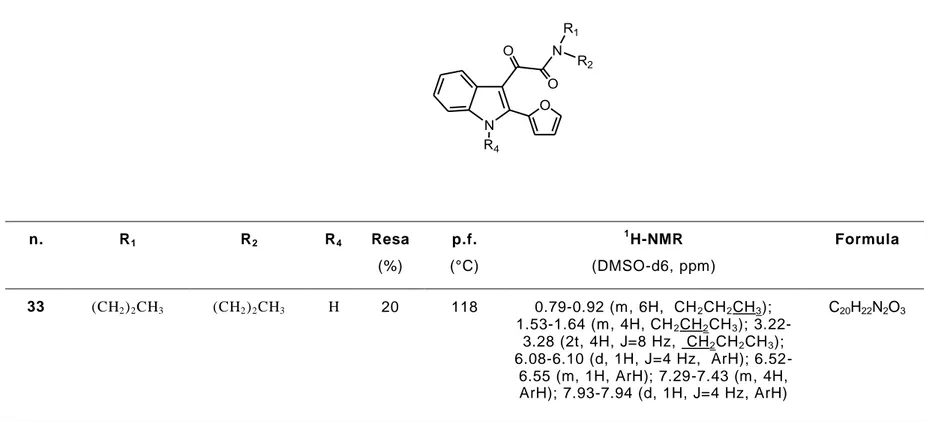
Tabella 3
. Costanti chimico-fisiche e caratteristiche analitiche e spettroscopiche dei derivati 33-34. N N O O R1 R2 R4 O n. R1 R2 R4 Resa (%) p.f. (°C) 1 H-NMR (DMSO-d6, ppm) Formula 33 (CH2)2CH3 (CH2)2CH3 H 20 118 0.79-0.92 (m, 6H, CH2CH2CH3); 1.53-1.64 (m, 4H, CH2CH2CH3); 3.22-3.28 (2t, 4H, J=8 Hz, CH2CH2CH3); 6.08-6.10 (d, 1H, J=4 Hz, ArH); 6.52-6.55 (m, 1H, ArH); 7.29-7.43 (m, 4H, ArH); 7.93-7.94 (d, 1H, J=4 Hz, ArH) C20H22N2O334 (CH2)2CH3 (CH2)3CH3 CH3 38 114-116 0.70-0.88 (m, 6H, CH2CH2CH3); 1.09-1.21 (m, 2H, CH2CH2CH3); 1.30-1.42 (m, 2H, CH2CH2CH3); 2.98-3.06 (2t, 4H, J=8 Hz, CH2CH2CH3); 3.50 (s, 3H, NCH3); 6.06-6.11 (d, 1H J=4 Hz, ArH); 6.50-6.56 (m, 1H, ArH); 7.10-7.29 (m, 4H, ArH); 7.80-7.83 (d, 1H, J=4 Hz ArH) C21H24N2O3