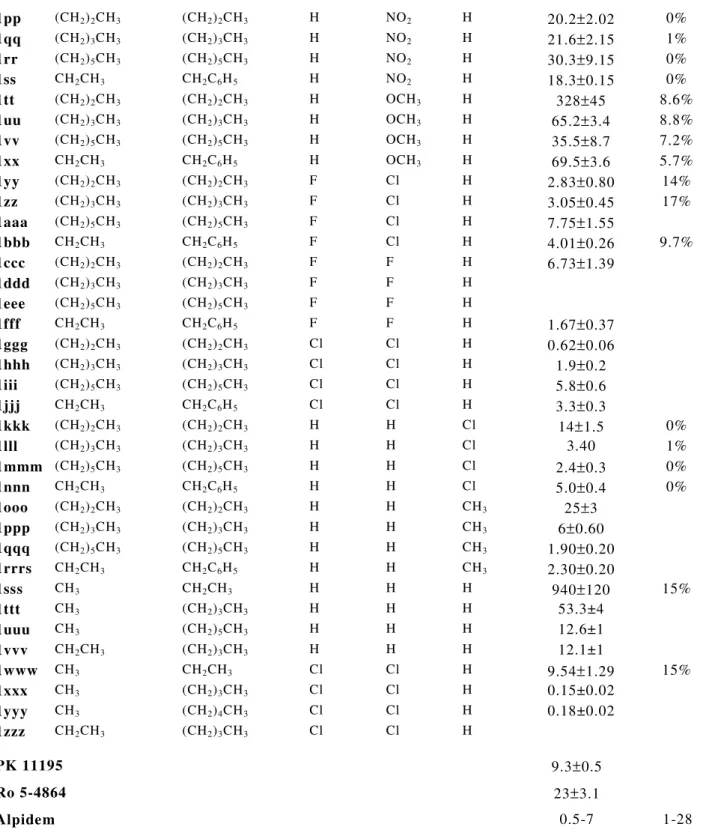
Tabella 1. Dati di binding dei derivati 2-arilindol-3-ilgliossilamidici 1a-zzz.
[6] N R1 R2 R3 R4 R5 PBR [a] Ki(nM) CBR [ b] 1a (CH2)2CH3 H H H H 815±80 3293±381 1b (CH2)3CH3 H H H H 1167±99 12% 1c CH2CH3 CH2CH3 H H H 43.0±4 - 1d (CH2)2CH3 (CH2)2CH3 H H H 12.2±1.0 16% 1e (CH2)3CH3 (CH2)3CH3 H H H 7.50±0.7 3% 1f (CH2)4CH3 (CH2)4CH3 H H H 16.0±2.0 3% 1g (CH2)5CH3 (CH2)5CH3 H H H 1.40±0.2 10% 1h CH(CH3)2 CH(CH3)2 H H H 103±9.0 - 1i CH(CH3)CH2CH3 CH(CH3)CH2CH3 H H H 17.0±2.0 5% 1j CH2CH3 CH2C6H5 H H H 11.0±1.0 - 1k -(CH2)4 H H H 2400±125 4% 1l -(CH2)5- H H H 665±30 3% 1m -(CH2)6- H H H 33.0±3.0 3% 1n (CH2)2CH3 (CH2)2CH3 F H H 4.28±0.32 4.3% 1o (CH2)3CH3 (CH2)3CH3 F H H 2.40±0.81 0% 1p (CH2)5CH3 (CH2)5CH3 F H H 0.37±0.13 0% 1q CH2CH3 CH2C6H5 F H H 1.68±0.12 10% 1r (CH2)2CH3 (CH2)2CH3 Cl H H 4.65±0.52 14% 1s (CH2)3CH3 (CH2)3CH3 Cl H H 1.00±0.27 0% 1t (CH2)5CH3 (CH2)5CH3 Cl H H 0.55±0.19 4.2% 1u CH2CH3 CH2C6H5 Cl H H 1.30±0.15 7.3% 1v (CH2)2CH3 (CH2)2CH3 NO2 H H 0.95±0.1 1w (CH2)3CH3 (CH2)3CH3 NO2 H H 0.23±0.07 1x (CH2)5CH3 (CH2)5CH3 NO2 H H 0.27±0.10 1y CH2CH3 CH2C6H5 NO2 H H 1z (CH2)2CH3 (CH2)2CH3 CF3 H H 1.69±0.2 1aa (CH2)3CH3 (CH2)3CH3 CF3 H H 1.16±0.1 1bb (CH2)5CH3 (CH2)5CH3 CF3 H H 1cc CH2CH3 CH2C6H5 CF3 H H 1dd (CH2)2CH3 (CH2)2CH3 CH3 H H 5.50±0.98 1ee (CH2)3CH3 (CH2)3CH3 CH3 H H 3.80±0.91 1ff (CH2)5CH3 (CH2)5CH3 CH3 H H 1.60±0.13 1gg CH2CH3 CH2C6H5 CH3 H H 1.33±0.2 1hh (CH2)2CH3 (CH2)2CH3 H F H 2.67±0.48 1ii (CH2)3CH3 (CH2)3CH3 H F H 4.00±0.15 1jj (CH2)5CH3 (CH2)5CH3 H F H 0.37±0.12 1kk CH2CH3 CH2C6H5 H F H 1ll (CH2)2CH3 (CH2)2CH3 H Cl H 2.80±0,3 1mm (CH2)3CH3 (CH2)3CH3 H Cl H 4.91±0.4 1nn (CH2)5CH3 (CH2)5CH3 H Cl H 58.4±6 3% 1oo CH2CH3 CH2C6H5 H Cl H 4.6±0,5continua
N N O O H R4 R1 R2 R3 R5Tabella 1. continua
1pp (CH2)2CH3 (CH2)2CH3 H NO2 H 20.2±2.02 0% 1qq (CH2)3CH3 (CH2)3CH3 H NO2 H 21.6±2.15 1% 1rr (CH2)5CH3 (CH2)5CH3 H NO2 H 30.3±9.15 0% 1ss CH2CH3 CH2C6H5 H NO2 H 18.3±0.15 0% 1tt (CH2)2CH3 (CH2)2CH3 H OCH3 H 328±45 8.6% 1uu (CH2)3CH3 (CH2)3CH3 H OCH3 H 65.2±3.4 8.8% 1vv (CH2)5CH3 (CH2)5CH3 H OCH3 H 35.5±8.7 7.2% 1xx CH2CH3 CH2C6H5 H OCH3 H 69.5±3.6 5.7% 1yy (CH2)2CH3 (CH2)2CH3 F Cl H 2.83±0.80 14% 1zz (CH2)3CH3 (CH2)3CH3 F Cl H 3.05±0.45 17% 1aaa (CH2)5CH3 (CH2)5CH3 F Cl H 7.75±1.55 1bbb CH2CH3 CH2C6H5 F Cl H 4.01±0.26 9.7% 1ccc (CH2)2CH3 (CH2)2CH3 F F H 6.73±1.39 1ddd (CH2)3CH3 (CH2)3CH3 F F H 1eee (CH2)5CH3 (CH2)5CH3 F F H 1fff CH2CH3 CH2C6H5 F F H 1.67±0.37 1ggg (CH2)2CH3 (CH2)2CH3 Cl Cl H 0.62±0.06 1hhh (CH2)3CH3 (CH2)3CH3 Cl Cl H 1.9±0.2 1iii (CH2)5CH3 (CH2)5CH3 Cl Cl H 5.8±0.6 1jjj CH2CH3 CH2C6H5 Cl Cl H 3.3±0.3 1kkk (CH2)2CH3 (CH2)2CH3 H H Cl 14±1.5 0% 1lll (CH2)3CH3 (CH2)3CH3 H H Cl 3.40 1% 1mmm (CH2)5CH3 (CH2)5CH3 H H Cl 2.4±0.3 0% 1nnn CH2CH3 CH2C6H5 H H Cl 5.0±0.4 0% 1ooo (CH2)2CH3 (CH2)2CH3 H H CH3 25±3 1ppp (CH2)3CH3 (CH2)3CH3 H H CH3 6±0.60 1qqq (CH2)5CH3 (CH2)5CH3 H H CH3 1.90±0.20 1rrrs CH2CH3 CH2C6H5 H H CH3 2.30±0.20 1sss CH3 CH2CH3 H H H 940±120 15% 1ttt CH3 (CH2)3CH3 H H H 53.3±4 1uuu CH3 (CH2)5CH3 H H H 12.6±1 1vvv CH2CH3 (CH2)3CH3 H H H 12.1±1 1www CH3 CH2CH3 Cl Cl H 9.54±1.29 15% 1xxx CH3 (CH2)3CH3 Cl Cl H 0.15±0.02 1yyy CH3 (CH2)4CH3 Cl Cl H 0.18±0.02 1zzz CH2CH3 (CH2)3CH3 Cl Cl H PK 111959.3±0.5 Ro 5-4864 23±3.1 Alpidem 0.5-7 1-28
[a] La concentrazione dei composti saggiati che inibisce il 50% del legame del [3H]PK11195 nelle membrane mitocondriali di rene di ratto (IC50) è stata determinata mediante analisi log-probit utilizzando 6 concentrazioni diverse del composto. I valori di ki sono medie ± SEM di 3 determinazioni. [b] Le percentuali di inbizione del legame specifico del [3H]Ro 15.1788 ad una concentrazione di 10µM del composto sono medie ± SEM di 5 determinazioni. I valori di ki sono medie ± SEM di 3 determinazioni.
![Tabella 1. Dati di binding dei derivati 2-arilindol-3-ilgliossilamidici 1a-zzz. [6] N R 1 R 2 R 3 R 4 R 5 PBR [a] K i (nM) CBR [ b] 1a (CH 2 ) 2 CH 3 H H H H 815±80 3293±381 1b (CH 2 ) 3 CH 3 H H H H 1167±99 12% 1c CH 2 CH 3 CH 2 CH 3 H](https://thumb-eu.123doks.com/thumbv2/123dokorg/7341273.91977/1.892.103.826.210.1151/tabella-dati-binding-derivati-arilindol-ilgliossilamidici-pbr-cbr.webp)